Computational Flow Dynamics of the Severe M1 Stenosis Before and After Stenting
Article information
Abstract
Purpose
Computational flow dynamic (CFD) study has not been widely applied in intracranial artery stenosis due to requirement of high resolution in identifying the small intracranial artery. We described a process in CFD study applied to symptomatic severe intracranial (M1) stenosis before and after stenting.
Materials and Methods
Reconstructed 3D angiography in STL format was transferred to Magics (Materialise NV, Leuven, Belgium) for smoothing of vessel surface and trimming of branch vessels and to HyperMesh (Altair Engineering Inc., Auckland, New Zealand) for generating tetra volume mesh from triangular surface-meshed 3D angiogram. Computational analysis of blood flow in the blood vessels was performed using the commercial finite element software ADINA Ver 8.5 (ADINA R & D, Inc., Lebanon, MA). The distribution of wall shear stress (WSS), peak velocity and pressure in a patient was analyzed before and after intracranial stenting.
Results
Computer simulation of wall shear stress, flow velocity and wall pressure before and after stenting could be demonstrated three dimensionally by video mode according to flow vs. time dimension. Such flow model was well correlated with angiographic finding related to maximum degree of stenosis. Change of WSS, peak velocity and pressure at the severe stenosis was demonstrated before and after stenting. There was no WSS after stenting in case without residual stenosis.
Conclusion
Our study revealed that CFD analysis before and after intracranial stenting was feasible despite of limited vessel wall dimension and could reveal change of WSS as well as flow velocity and wall pressure.
Because intracranial atherosclerotic stenosis is angiographically more common than extracranial lesions in Koreans, intracranial stenting has been performed more frequently than in Western countries (1-3). Although intracranial angioplasty and/or stenting is believed to be beneficial for patients who are resistant to medical treatment, appropriate indication of intracranial stenting and long term outcome data is still not sufficient (4-6).
Although final luminal diameter after stenting is critical for the patient outcome, hemodynamic factors prone to adverse events or restenosis leading to target vessel revascularization in intracranial artery have not been well demonstrated (4). Few studies of the hemodynamics in small-caliber intracranial vessels have been conducted especially in vessels associated with severe stenosis (5-9). The reason of the difficulties to apply this computational flow dynamics (CFD) research to intracranial artery is in part due to the limited resolution of stenotic lumen imaged by current technologies because resolution of target vessel has precluded the development of a realistic geometry for use in finite element modeling and CFD analysis (10, 11).
We developed a process of CFD technique to elucidate the local hemodynamics as a result of atherosclerosis of the small intracranial arteries before and after intracranial stenting.
MATERIALS AND METHODS
Three D Reconstruction and Data Transfer
Three dimentional (3D) rotational angiography was obtained in AXIOM Artis Zee (Siemens, Erlangen, Germany). The 3D angiographic images were transferred to Syngo workstation (version VB 15D) to generate 3D angiography from which 2nd reconstruction was performed by reducing FOV and adjusting the target vessel. The final image was transformed to STL format in 256×256 pixels. Because there was no direct connection between the Syngo workstation and the images were sent to Magic RP (ver. 12. 01, Materialise, Leuven, Belgium) in other workstation for smoothing of vessel surface and trimming of branch vessels. And then the image tranfered to Hypermesh (Altair Engineering, Inc., Auckland, New Zealand) to generate tetra volume mesh from triangular surface-meshed 3D angiogram.
CFD Analysis
Computational analysis of blood flow in the blood vessels was performed using the commercial finite element software ADINA Ver 8.5 (ADINA R & D, Inc., Lebanon, Massachusetts, USA). Blood flow was assumed to be laminar, viscous, Newtonian, and incompressible because of its inherent flow characteristics. No-slip boundary conditions were assumed for the flow viscosity produced between the fluid and the wall surface of the blood vessels. Simulations were performed with the following material constants: blood density, 1100 kg/m3, and blood dynamic viscosity, 0.004 Poiseuille. To achieve truly patient-specific modeling, the boundary conditions at the inflow boundary were based on the pulsatile periodic flow rate. Maximum wall shear stress (WSS) and flow velocities before and after stenting were obtained.
The velocity and flow rate of the internal carotid artery were obtained from gated phase contrast MR angiography in an age-matched male who did not have any intracranial vascular disease. The average velocities in the systolic and diastolic phases were 0.45 m/s and 0.23 m/s, respectively.
Patient Data
A 65-year-old male patient who presented with right arm weakness revealed an acute focal cortical ischemic change in the borderzone type on the diffusion-weighted image (not shown). He had diabetes mellitus and history of alcohol. He did not have arterial fibrillation or any coagulation disorders. Our Institutional Review Board approved this study, and we obtained written informed consent from the patient and the patient's family.
RESULTS
Cerebral angiogram after stenting showed no residual stenosis of the right M1 segment and the shift of borderzone between the anterior and the middle cerebral artery territory toward normal position (5, 6). Maximum WSS before and after stenting were shown in Figure 1C and D. Maximum WSSs before stenting were 18.5 Pa in systole and 9.3 Pa in end-diastole and those after stenting were 7.1 Pa in systole and 3.6 Pa. Maximum flow velocities before stenting were 135 cm/s in systole and 70 cm/s in diastole and those after stenting were 72 cm/s in systole and 38 cm/s in diastole.
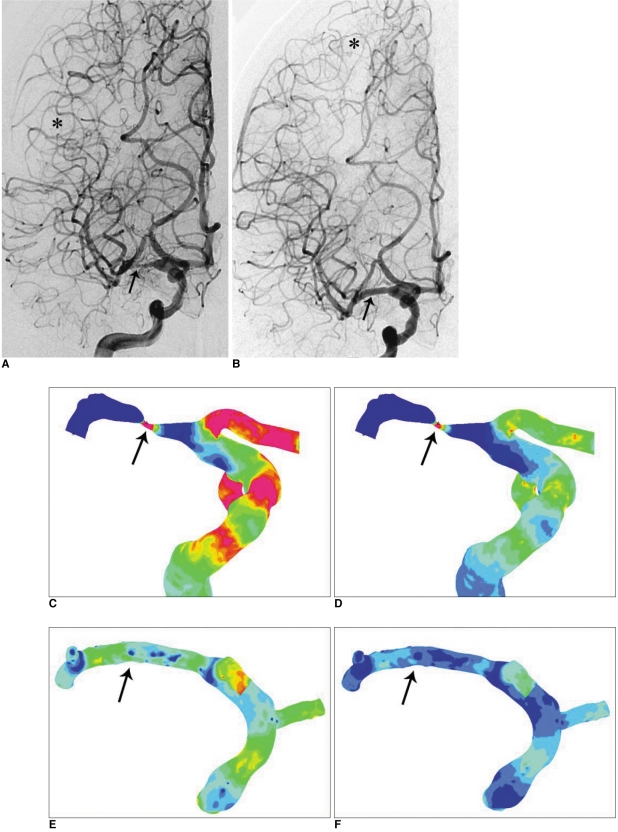
Cerebral angiogram and Computational flow dynamics (CFD) study in the severe M1 stenosis. Angiograms before (A) and after (B) stenting show relieved severe stenosis of the right M1 and normalization of decreased size and filling of the right MCA branches after stenting (arrows in A & B). Note upward shift of borderzone area to the upward normal postion after stenting (asterisk in B compared to A).
C, D. Contour plot of the wall shear stress (WSS) map before stenting shows an increased WSS corresponding to the stenosis site during systole (C) and end-diastole (D) (arrows).
E, F. Contour plot of the WSS map after stenting shows disappearance of high WSS area at normalized contour of the right M1suggesting hemodynamically successful stenting during systole (E) and end-diastole (F) (arrows).
Compared to previously reported process of CFD analysis, we could omit the several steps reduce the time spending in data processing (11, 12). Smoothing of vessel surface and trimming of branch vessels before CFD analysis required still considerable time to generate tetra volume mesh.
DISCUSSION
Since we demonstrated that CFD analysis of the small-caliber intracranial artery was feasible and could be correlated with the atherosclerotic plaque in the stenotic segment, we obtained CFD before and after intracranial stenting and further simplified the process which could reduce analysis time (11, 12). Disappearance of WSS after stenting can be used as a beneficial landmark in assessing appropriate final luminal diameter preventing further angioplasty and even may predict minimal chance of restenosis at the hemodynamical point of view although follow-up study is mandatory to reach such a conclusion (12-14).
We found that the smoothing and trimming of branch vessels of the model vessel in Magic RP required a compromise. If we get high resolution image, it will takes time to generate too much tetra volume mesh. If we get low resolution image, processing time will be reduced but identification or differentiation of parent or branch vessels will be difficult (11).
Our study has several limitations. Firstly, image transfer from 3D angiogram to ADINA software was not yet possible and thus required a time-consuming tetra volume mesh generation even though we could reduce processing time since we used ADINA. If 3D angiogram is directly transferred to CFD analysis software such as ADINA, CFD analysis will be more readily applicable. Secondly, flow data in each patient was not readily provided for boundary condition in inflow as well as outflow zones of the model vessel (11, 15). Normal standard value of flow velocity according to age and gender groups might be helpful for the general usage. In addition, flow data from transcranial Doppler or MR phase study can be measured and used in each patient (10, 11).
In conclusions, we could reduce CFD processing time compared to previous report. We also demonstrated change of WSS, flow velocity and wall pressure in the severe stenotic intracranial artery, that is, M1 before and after stenting. This observation can provide a predictable risk factors affecting patient's outcome if the study can match to the long term follow-up data.
Acknowledgements and Funding
This study was supported by a grant from the Korea Healthcare Technology R&D Project, Ministry of Health & Welfare, Republic of Korea. (A080201)