Endovascular Treatment of Symptomatic Basilar Artery Stenosis
Article information
Abstract
Purpose
While symptomatic basilar artery (BA) stenosis is associated with a higher risk of recurrent stroke or death, there is no consensus on the management of these patients who are refractory to antiplatelet therapy. This study retrospectively assesses the outcomes of endovascular treatment (EVT) for symptomatic BA stenosis.
Materials and Methods
We conducted a retrospective review of patients with symptomatic BA stenosis who underwent EVT, including angioplasty or stenting, from 2006 to 2018. A total of 15 patients, who experienced transient ischemic attacks or strokes despite dual antiplatelet therapy, were included. EVT was performed under local anesthesia after pretreatment with antiplatelet medications. Angiographic follow-up was performed at 12 and 24 months post-EVT. Clinical outcomes were evaluated using the modified Rankin Scale (mRS).
Results
EVT was successfully completed in all patients. Peri/post-procedural complications occurred in 33% of cases, including in-stent thrombosis, intracranial hemorrhage, and pontine infarction. At long-term follow-up (mean 98.5±80.5 months), 73.3% of patients achieved a favorable functional outcome (mRS≤2) without disability or mortality. Patients with unfavorable outcomes had previous infarcts, with 2 experiencing new pontine infarctions after stenting.
Conclusion
This study suggests that EVT, including angioplasty and stenting, may offer promise as a treatment option for symptomatic BA stenosis refractory to medical therapy. However, the procedure carries a notable risk of complications, especially in patients with severe stenosis and previous infarcts. Careful patient selection, based on clinical and radiological criteria, is crucial.
INTRODUCTION
Intracranial atherosclerotic stenosis (ICAS) is one of the leading causes of stroke worldwide and its prevalence is higher in Asia, accounting for more than 40% of ischemic strokes [1,2]. Unlike the anterior circulation, atherosclerosis is the major cause of stroke in the posterior circulation [3]. Although symptomatic basilar artery (BA) stenosis was rare, BA stenosis was highly associated with the risk of transient ischemic attack (TIA), stroke or death [4,5]. In addition, atherosclerotic BA stenosis is difficult to treat due to its eloquent location and the presence of multiple perforators.
In the Stenting and Aggressive Medical Management for Preventing Recurrent stroke in Intracranial Stenosis (SAMMPRIS) trial [6,7], patients with BA stenosis accounted for 21.9% of the intervention group and BA stenting had a higher risk of peri-procedural ischemic stroke (20.8% for BA vs. 6.7% for non-BA). There is no consensus regarding the management of patients with ICAS, and the individual approach would be tailored to the specific vascular anatomy and pathology of ICAS. However, patients with symptomatic ICAS who fail antithrombotic therapy have extremely high rates of recurrent TIA/stroke or death [8]. Therefore, endovascular treatment (EVT) may be a last resort for patients with symptomatic BA stenosis who are refractory to dual antiplatelet therapy.
For the management of symptomatic BA stenosis, we retrospectively reviewed our experience with patients who underwent BA stenting or angioplasty and tried to analyze the parameters associated with the clinical outcome during the long-term follow-up.
MATERIALS AND METHODS
Patients
The institutional review board approved this retrospective study, and informed consent was waived. A clinical database of patients with symptomatic BA stenosis who underwent EVT, including angioplasty or stenting, at our hospital from 2006 to 2018 was retrospectively reviewed. The exclusion criteria were acute BA occlusion due to thrombus or dissection, inability of angiography due to decreased renal function and contrast allergy.
Endovascular Procedure
All patients underwent diagnostic cerebral angiography prior to EVT to evaluate the associated vascular anatomy and lesion characteristics in the BA. Treatment decisions were made in consultation with neurologists and interventional neuroradiologists.
All patients were pretreated with antiplatelet therapy (clopidogrel, 75 mg/d and aspirin, 325 mg/d) 3 days before EVT. Intravenous heparin (activated clotting time >300) was administered during EVT and maintained for 24 hours after the procedure.
EVT was performed under local anesthesia in all patients. A 6F guide catheter (Envoy; Cerenovus) was placed into the vertebral artery via the femoral artery, and the stenotic lesion was crossed with a 0.014-inch microwire (Synchro 14 or Transend 300 guidewire; Stryker) and a balloon-expandable stent (BES, Flexmaster or Vision; Abbott) or self-expandable stent (Wingspan; Stryker) was deployed in the BA with or without angioplasty (Gateway balloon catheter; Stryker).
Clinical Outcome and Follow-up
All patients were evaluated by post-procedural angiography to confirm patency of the BA after stenting or angioplasty. Peri-procedural and postoperative complications, including in-stent thrombosis, reocclusion, dissection, vessel rupture, intracranial hemorrhage (ICH), subarachnoid hemorrhage, and embolic infarction, were also evaluated. Follow-up angiography was performed 12, 24 months after EVT to confirm restenosis or occlusion of the BA. Computed tomographic angiography was used as an alternative for follow-up when digital subtraction angiography (DSA) was not available. The modified Rankin Scale (mRS) was used to assess functional outcome at discharge and at 3-month follow-up.
RESULTS
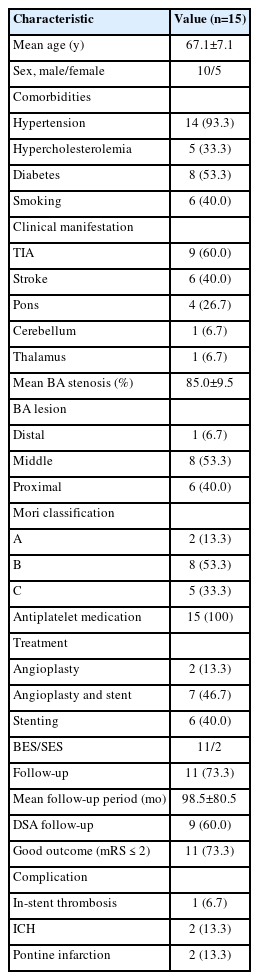
Of 22 patients, 6 cases with BA occlusion and 1 case with BA dissection were excluded. Fifteen patients who underwent percutaneous transluminal angioplasty (PTA) and stenting for treatment of symptomatic BA stenosis were enrolled (Table 1). All patients experienced TIA or stroke despite a dual antiplatelet therapy. The average stenosis of the BA was more than 85.0%, and the stenotic lesion was predominantly located in the middle BA (8, 53.3%) and proximal BA (6, 40.0%). According to the Mori classification [9], there were 8 cases of type B, 5 cases of type C and 2 cases of type A.
EVT was successfully performed in all patients, including 2 cases treated with angioplasty alone, 7 cases with PTA and stenting, and 6 cases with primary stenting. BESs were used in most cases, while wingspan stents were used in 2 cases. Peri-procedural or post-procedural complications were instent thrombosis in 1 patient, ICH in 2, and pontine infarction in 2. Except for pontine infarction, patients with ICH did not develop neurological deterioration after the procedure, and intra-arterial thrombolysis with tirofiban (1 mg) was performed in 1 patient with in-stent thrombosis.
Of the 15 patients, 9 were available for angiographic follow-up (Fig. 1). The mean follow-up was 98.5±80.5 months. At long-term follow-up, 11 patients (73.3%) had a good functional outcome (mRS≤2) without in-stent stenosis or BA occlusion.

Symptomatic stenosis of the mid-basilar artery. (A) Prestenting, (B) post-stenting, and (C) 1 year follow-up angiographic images.
While 4 patients with an unfavorable outcome were treated with dual antiplatelet therapy more than 3 days before procedures, fluctuation of their neurological symptoms was developed. Three patients had a previous infarct involving the pons and thalamus at admission (Table 2). BESs were used in 4 patients. After stenting, 2 patients had a new pontine infarct due to perforator occlusion and became vegetative with neurological symptoms worse than before admission (mRS=5), and 1 patient with a previous bilateral pontine infarct (mRS=4) and 1 patient with a previous thalamic infarct (mRS=3) were discharged from the hospital without neurological improvement (Fig. 2).
DISCUSSION
In contrast to the anterior circulation stroke, 60% of posterior circulation strokes are due to atherosclerosis [3] and the prevalence of isolated symptomatic BA stenosis has been reported to be 1.43% [10]. Although BA stenosis is rare, symptomatic BA stenosis is a strong independent predictor of stroke recurrence [11]. Samaniego et al. [10] proposed that mid-BA stenosis, ≥80% stenosis, and poor collateral circulation were significantly associated with poor prognosis. In this study, we demonstrated that EVT with stenting or angioplasty may be a promising option for the treatment of symptomatic BA stenosis, with a high success rate, and a low rate of instent restenosis and recurrent stroke at long-term follow-up. However, the peri/post-procedural complication rate was 33% (5/15), which were higher than the similar reports [12-15]. Although ICH or in-stent thrombosis was asymptomatic, 2 pontine infarctions were fatal. The cause of the high procedural complication may be explained by the fact that most of the stenotic lesions were mid-BA, severe stenosis of more than 80%, frequent use of BES, and the presence of previous infarction involving the pons or thalamus on the initial magnetic resonance imaging (MRI). Luo et al. [16] reported that high burden plaques located in the middle segment and lateral wall of the BA are likely to increase postoperative complications after EVT. In addition, BA plaques have a concentric distribution, a higher incidence of intraplaque hemorrhage, a higher proportion of internal elastic lamina and less elastin than the anterior circulation [17-19].
The current treatment options for BA stenosis are (1) aggressive medical therapy, including antiplatelet therapy and strict control of risk factors, and (2) EVT, including angioplasty and stenting [20]. Although Jiang et al. [21] reported that a significantly higher risk of stroke after stenting exists in patients with severe BA stenosis, Palmisciano et al. [22] suggested that elective angioplasty and/or stenting appears to be safe and effective in selected patients with medically refractory, severe, symptomatic, and non-acute BA stenosis. In this meta-analysis study of 1,016 patients from 25 retrospective cohort studies, the actuarial rates of successful intervention and good outcome were 100% and 89%, respectively. Intervention-related recurrent ischemic stroke and death occurred in 85 patients (8.3%) and 0%, respectively. Recently, some studies have been published to improve the safety of EVT for symptomatic BA stenosis by analyzing the plaque distribution by MR vessel wall imaging or measuring the hemodynamic change after stenting by quantitative DSA to predict the prognosis of patients after EVT [16,23]. Therefore, it is necessary to carefully select patients based on clinical and radiological decision and actively consider EVT for these selected patients.
This study has some limitations in that it was a retrospective design with small number of enrolled cases and ethnic bias, which makes the possibility of selection bias. More prospective, randomized clinical trials are needed to resolve the controversies and debate for better clinical outcome.
CONCLUSION
In management of symptomatic BA stenosis, EVT with stenting or angioplasty provides another option in patients who are refractory to aggressive medical therapy. While the rate of peri/post-procedural complication is still high, careful patient selection is crucial according to clinical and radiological decision. Further randomized studies are necessary in determining the safety and efficacy of EVT for symptomatic BA stenosis for selected patients.
Notes
Fund
None.
Ethics Statement
The ethics approval was obtained from the institutional review board and the informed consent was waived. This article does not include any information such as age and sex that may identify the person.
Conflicts of Interest
SHS has been the Editor-in-Chief of the Neurointervention since 2023; however, SHS has not been involved in the peer reviewer selection, evaluation, or decision process of this article. No potential conflict of interest relevant to this article was reported. No other authors have any conflict of interest to disclose.
Author Contributions
Concept and design: SHS. Analysis and interpretation: KCC, TK, and SWH. Data collection: JHK, KCC, and TK. Writing the article: JHK and SHS. Critical revision of the article: KCC, SWH, and SHS. Final approval of the article: SHS. Statistical analysis: JHK and TK. Overall responsibility: SHS.