Intracranial Stenting; the Current Landscape
Article information
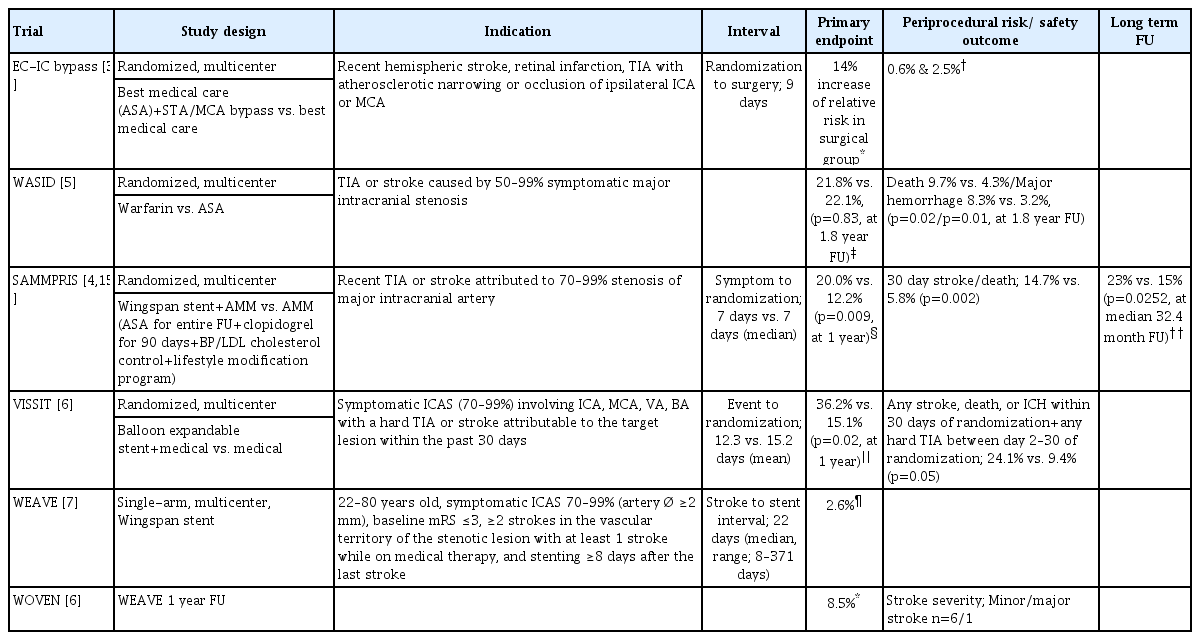
Intracranial atherosclerotic stenosis (ICAS) is one of the most common causes of stroke worldwide, and it is of special concern in the Asian population due to its higher prevalence [1,2]. ICAS is the cause of ischemic stroke in 30–50% in Asians, compared to 8–10% in North America [2]. The high recurrence rate and the disability caused by the stroke warrants a safe and effective treatment method. Bypass surgery as well as endovascular treatment have been tested against best the medical therapy, but current best evidence does not support either surgery or intervention as the primary treatment modality for ICAS lesions (Table 1) [3-6].
In this regard, the Stenting and Aggressive Medical Management for Preventing Recurrent Stroke in Intracranial Stenosis (SAMMPRIS) randomized trial and its post-hoc studies in addition to the single-arm Wingspan Stent System Post Market Surveillance (WEAVE) trial and its 1 year follow up results (Wingspan One-year Vascular Events and Neurologic Outcomes [WOVEN] trial) published last year shed some insights into the importance of patient selection concerning who may or may not benefit from intracranial stenting for ICAS lesions [4,7,8].
The WEAVE trial adhered strictly to the US Food and Drug Administration (FDA) on label usage criteria in terms of patient age, stenosis degree, baseline neurological status, type of stroke, medical failure, and time to intervention, resulting in a highly selected group of patients for stenting (Table 1) [7]. The results of the 1-year follow-up of the WEAVE trial (WOVEN trial) showed a primary endpoint rate of 8.5%. Postperiprocedural (1 month to 1 year) event rates were comparable between the WOVEN (5.4%) vs. SAMMPRIS stenting (5.3%) vs. SAMMPRIS aggressive medical management (AMM, 6.5%) results. Moreover, most of the strokes in the postperiprocedural period were minor strokes (6 of 7 cases) suggesting that decreasing the periprocedural event rate is critical and that there may be clinical benefits of stenting at 1 year follow up in these selected patients [7,8]. This is uplifting news; however, good single-arm interventional results have not been duplicated in double-arm studies for ICAS. Further trials comparing these best stenting protocols with the best medical therapy are warranted.
Another important insight is that the pathological mechanism of stroke should be distinguished. There is growing evidence that patients with hemodynamic dominant stroke may benefit most in contrast to those with perforator stroke. Although most subgroup analyses of the SAMMPRIS study did not show favorable results for the stenting arm, the post-hoc analysis of the medical arm showed that recurrent stroke rates were higher in patients with a borderzone infarct pattern on magnetic resonance imaging/computed tomography and impaired collateral flow (36.7%) compared to other groups combined (7.7%) (p<0.05). The primary endpoint rates were numerically lower in the stenting arm in patients with hemodynamic infarct compared to the AMM arm (18.2% vs. 26.4%), suggesting that these patients may be a promising population for stenting [9]. The WEAVE trial did not systematically exclude patients with perforator-type stroke; however, underdilation of the balloon to 60% rather than the standard 80% of the normal vessel diameter was recommended when directly adjacent to visible perforators. The resulting rate of perforator stroke in this trial was only 0.7% [7].
Issues remain despite these promising new insights. Strict adherence to the conservative FDA indications for ICAS stenting may result in super selection of patients [10]. Many aspects, including the limitation of patients to medical failure (≥2 strokes with at least 1 stroke while on medical therapy), type of qualifying event (stroke or transient ischemic attack), and the time to treatment, may need further investigation [11-14]. In terms of time to treatment, the current major focus in ICAS stenting is decreasing the high periprocedural risk. Thus, stenting is often delayed to avoid risks associated with unstable plaque and possibly hyperperfusion [15,16]. However, the recurrent stroke rate is highest in this early post-ictus period; thus, an effective treatment should target this critical period. Lessons learned from recent studies and the ongoing growth of experience in areas, such as rescue intracranial stenting in ICAS related acute stroke, may interplay to open new doors in the near future [17].
Meanwhile, we await newer insights from the Angioplasty and Stenting for Patients With Symptomatic Intracranial Atherosclerosis (NCT02689037) and the China Angioplasty & Stenting for Symptomatic Intracranial Severe Stenosis (CASSISS, NCT01763320) trials [18,19].
Notes
Fund
None.
Conflicts of Interest
DJK has been the Associate Editor of the Neurointervention since 2018. No potential conflict of interest relevant to this article was reported.