Mechanical Thrombectomy for Septic Embolism Secondary to Staphylococcus lugdunensis Bacteremia without Infective Endocarditis: A Case Report
Article information
Abstract
A cerebral large vessel occlusion due to septic embolism with resultant stroke is a known complication of infective endocarditis and can cause severe neurologic disability. However, septic embolism rarely occurs in the absence of infective endocarditis, and emboli due to different organisms may behave differently. As such, it is important to recognize the different pathogens that can cause septic embolism resulting in cerebral large vessel occlusion and to have data on successful treatments. We describe here a case of mechanical thrombectomy for septic embolism secondary to Staphylococcus lugdunensis bacteremia without infective endocarditis.
INTRODUCTION
Ischemic stroke occurs in an estimated 40% of patients with infective endocarditis (IE) and is associated with a higher morbidity and mortality [1]. In patients with active IE who are found to have emergent large vessel occlusions (ELVO), the Society recommends that mechanical thrombectomy (MT) be considered as the risks and benefits are similar compared to MT in patients without IE [2]. IE due to Staphylococcus lugdunensis (S. lugdunensis) is associated with a high mortality rate, especially in patients who are only treated medically [3,4]. We report here on a case of successful MT for ELVO due to an S. lugdunensis septic embolism in a patient with bacteremia in the absence of endocarditis.
CASE REPORT
An early geriatric-aged patient with a medical history of human immunodeficiency virus (HIV) with a most recent cluster of differentiation 4 (CD4) cell count of 44/mm3, hepatitis C, and liver cirrhosis presented to the emergency department with back pain and urinary hesitancy. The patient was diagnosed with presumed cystitis and admitted for infectious work-up including blood and urine cultures. Sulfamethoxazole-trimethoprim and broad-spectrum antibiotics were initiated for Pneumocystis prophylaxis and blood culture gram stains positive for gram-positive cocci in clusters, respectively. A transesophageal echocardiogram was recommended to rule out endocarditis, but the patient declined the procedure.
On hospital day 3, the patient was found to have right-sided hemiparesis, aphasia, and a leftward gaze with a last known normal of 3 hours prior. The National Institutes of Health stroke scale was 14, and computed tomography (CT) angiography and perfusion revealed an occlusion at the left M1/M2 junction of the middle cerebral artery (MCA) with a core infarct of 20 mL and penumbra of 144 mL. Tissue plasminogen activator was not given as there was concern for septic emboli from presumed endocarditis.
Under general anesthesia, the right common femoral artery was accessed, and an 8-F Endophys Pressure Sensing Access Sheath (Endophys, Dallas, TX, USA) was placed. A 4-F Angled Hydrophilic Coated Glidecath (Terumo Medical, Tokyo, Japan) was advanced through the sheath over a 0.035-inch × 180-cm Angled Tip Guidewire (Terumo Medical) to the origin of the left common carotid artery. Digital subtraction angiography (DSA) confirmed an occlusion of the left distal MCA trunk (Fig. 1A).
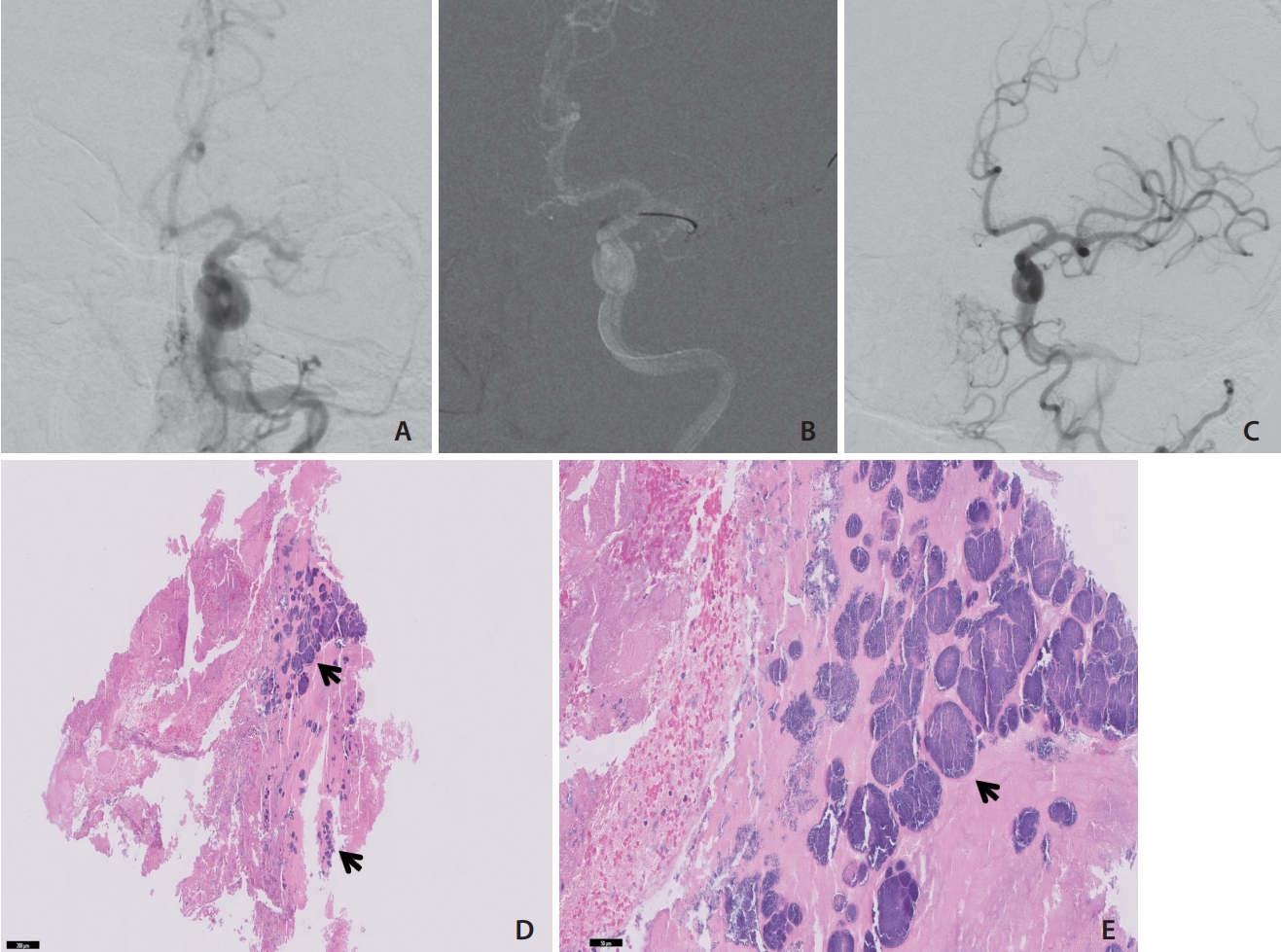
Diagnostic cerebral angiogram demonstrating left middle cerebral artery occlusion in the anterior-posterior (AP) view (A). AP road map (B) demonstrates deployment of the EMBOTRAP (Cerenovus, Miami, FL, USA) across the embolus. An AP view angiogram (C) demonstrates modified thrombolysis-in-cerebral-infarction 2C reperfusion after embolectomy. H&E stains under 5× (D) and 20× (E) magnification demonstrate an embolism with bacterial colonization (blue arrows).
Over a 0.035-inch × 260-cm exchange length Glidewire (Terumo Medical), the Glidecath was replaced with a 132-cm large bore catheter (LBC) (Cerenovus, Miami, FL, USA) inside an 8-F Cerebase DA Guide Sheath (Cerenovus). The exchange length Glidewire was removed. A combination Synchro-14 Guidewire (Stryker, Kalamazoo, MI, USA) in a Phenom 27 Catheter (Medtronic, Dublin, Ireland) was advanced through the LBC, and the Phenom was advanced through the occlusion to an M3 division of the MCA. Suction pump aspiration (in a manner standard for direct aspiration thrombectomy) was initiated through the LBC, and an EMBOTRAP III retrievable stent (Cerenovus) was delivered through the Phenom and unsheathed within M1/M2 (Fig. 1B). The EMBOTRAP was then withdrawn into the LBC and the LBC was withdrawn. As these were withdrawn, we initiated simultaneous syringe aspiration of the Cerebase DA catheter with a 60 mL syringe to capture any free emboli that could migrate during EMBOTRAP/LBC withdrawal. Three pale white emboli were aspirated and sent to pathology. DSA showed a modified thrombolysis in cerebral infarction (mTICI) score of 2C (Fig. 1C). Magnetic resonance imaging of the brain the next day demonstrated small, scattered infarcts in the distribution of the left MCA without significant mass effect or hemorrhage.
Histopathologic examination of the embolus demonstrated the presence of gram-positive cocci within the embolus (Fig. 1D, E). The patient’s blood cultures and urine culture, which were collected upon admission, and the intraoperative embolic tissue cultures were all positive for S. lugdunensis. The patient underwent a transthoracic echocardiogram the day after MT, which did not demonstrate any valvular vegetations. The patient then underwent a transesophageal echocardiogram for further work-up, but neither evidence of endocarditis nor an intracardiac shunt was identified. It did, however, demonstrate findings suggestive of an intrapulmonary shunt. Other imaging, including positron emission tomography and contrast-enhanced CT of the chest, abdomen, and pelvis, demonstrated lumbar discitis, possible colitis, possible pancreatitis, and possible duodenitis. The patient’s neurologic deficits resolved over the course of the hospital stay, and the patient was discharged to a skilled nursing facility for 6 weeks of intravenous nafcillin therapy. It was noted on a therapy evaluation just prior to discharge that the patient’s modified Rankin score was 2.
DISCUSSION
Septic embolism is a broad term that describes an embolus, often originating from a thrombus or IE vegetation, colonized by bacteria [5]. Septic emboli can obstruct any blood vessel in the body and result in devastating immediate and delayed sequelae [5,6]. The treatment paradigm includes both antibiotics and surgery of the source or affected tissue (such as valve replacement, percutaneous drainage, or splenectomy). Blood cultures often reveal the causative organism, but tissue sampling is sometimes required for a definitive diagnosis, and polymicrobial infections are possible. For immunocompromised patients, such as those with HIV, there is additionally a need to consider atypical infections. Our patient had both HIV and cirrhosis, and a thorough infectious work-up was initiated. He underwent extended antibiotic treatment and successful thrombectomy for cerebral ELVO due to S. lugdunensis septic embolism in the absence of endocarditis. In the literature, there are only 3 other such cases: 1 left ventricular assist device-related, 1 pneumonia-related, and 1 urinary tract infection-related [7,8]. Thus, the frequency of patients without endocarditis undergoing MT for cerebral septic embolism is exceedingly rare, and none of these patients were positive for S. lugdunensis.
S. lugdunensis is a commensal coagulase-negative Staphylococcus species that makes up part of the skin flora. Although often harmless, S. lugdunensis can be associated with a relatively high morbidity and mortality [4,9]. Two cases of S. lugdunensis septic embolism in the absence of IE have been reported [10,11]. Ishidou et al. [10] reported a patient underwent prolonged antibiotic therapy for a month but decompensated a month after discontinuation, and the conduit, a polytetrafluoroethylene foreign body, was found to be obstructed during autopsy. Shah et al. [11] described the patient underwent multiple antibiotic courses for recurrent bacteremia and finally underwent surgical ligation of a coronary artery fistula, which was presumed to be the source of bacteremia, without any further recurrence. Both cases illustrate the importance of a combination of medical and surgical intervention when appropriate under close monitoring in patients with S. lugdunensis bacteremia, even in the absence of IE. Although thrombectomy was performed in our patient, no other surgical procedure was performed as no embolic source was identified.
This case suggests that MT utilizing combination aspiration-stent retrieval may be an effective treatment for patients with ELVO due to septic embolism. Marnat et al. [12] reported that 28 patients with IE (compared to other patients with cerebral thromboembolism) had poorer 90-day functional outcomes but had similar rates of successful thrombectomy, postoperative hemorrhage, and mortality. In a larger propensity-matched German cohort including 55 patients with IE, the mortality rate of patients with IE was nearly double [13]. Further studies are needed to elucidate whether septic emboli due to different pathogens behave differently and thus would be optimally treated differently. This hypothesis is supported by the differing virulence and vegetation sizes associated with IE due to different organisms, which is thought to be related to bacterial upregulation of the platelet and coagulation cascade [14].
Herein, We demonstrated the rare case of a S. lugdunensis infection resulting in bacteremia and MCA occlusion that was successfully treated by MT with a stent retriever and The endovascular treatment may be an effective treatment for patients with cerebral septic emboli.
Notes
Fund
None.
Ethics Statement
The Institutional Review Boards of Baylor Scott and White Health and Texas A&M University have waived the requirement for informed consent. The patient information has been anonymized.
Conflicts of Interest
The authors hereby declare that they have no relevant conflicts of interest to disclose. WSL is an investigator in the Medtronic (Dublin, Ireland) Embolization of the Middle Meningeal Artery With ONYX™ Liquid Embolic System for Subacute and Chronic Subdural Hematoma (EMBOLISE), but this patient was not part of the trial.
Author Contributions
Concept and design: AVPN, SRD, BL, and WSL. Analysis and interpretation: AVPN, SRD, BL, and WSL. Data collection: AVPN, SRD, BL, and WSL. Writing the article: AVPN, BL, and WSL. Critical revision of the article: AVPN, SRD, BL, and WSL. Final approval of the article: AVPN, SRD, BL, and WSL. Overall responsibility: WSL.
