Initial Experience with a New Self-Expanding Open-Cell Stent System with Antithrombotic Hydrophilic Polymer Coating (pEGASUS Stent) in the Treatment of Wide-Necked Intracranial Aneurysms
Article information
Abstract
Purpose
We report our initial experience with endovascular embolization of intracranial aneurysms using this new self-expanding open-cell stent system (pEGASUS stent system) with the antithrombogenic hydrophilic polymer coating.
Materials and Methods
We retrospectively reviewed all patients treated with stent-assisted coiling or the Woven EndoBridge device using the pEGASUS stent system between September 2022 and June 2023. Demographic, clinical, and angiographic data were analyzed as well as short-term follow-up, including procedural complication rates and aneurysmal occlusion rates using the Raymond–Roy occlusion classification (RROC).
Results
Twelve patients with 12 wide-necked intracranial aneurysms were treated with the pEGASUS stent system, including 2 acutely ruptured aneurysms embolized in an emergency setting. The treated aneurysms were located at the anterior communicating artery (25.0%), the basilar artery (50.0%), the middle cerebral artery (16.7%), and the internal carotid artery (8.3%). All stents were deployed successfully. Immediate complete aneurysmal occlusion (RROC class I) was achieved in 83.3% (10/12) and near-complete occlusion (RROC II) in 16.7% (2/12). No periprocedural complications occurred in patients treated in the elective setting. A single case of intraoperative in-stent thrombus formation occurred during the treatment of an acutely ruptured basilar aneurysm and was resolved with intravenous Tirofiban. No other periprocedural complications occurred. Eleven out of 12 patients were available for follow up (mean 7.4 months). Complete aneurysmal occlusion without in-stent stenosis (ISS) was seen in 10 patients (90.9%). One patient (9.1%) showed aneurysmal reperfusion (RROC IIIb) with asymptomatic moderate ISS.
Conclusion
Our initial results demonstrate that the pEGASUS stent system appears to be a safe and effective device for stent assisted embolization of wide-necked intracranial aneurysms. More data is necessary to evaluate long-term follow-up.
INTRODUCTION
The ongoing evolution of novel techniques and devices such as stent-assisted coiling (SAC), flow diverters, and intrasaccular devices has enabled endovascular treatment of complex intracranial aneurysms that were previously deemed uncoilable, such as wide-necked bifurcation aneurysms or aneurysms with an unfavorable dome to neck ratio [1-3]. The self-expanding stent systems used for SAC include both closed- and open-cell systems with a wide array of lengths and diameters. Compared to simple coiling, SAC reduces the aneurysmal recurrence rate allowing a higher density of coils while protecting the parent vessel from coil protrusion [4]. In addition, a change in arterial geometry by the deployed stent can change the flow vector in the parent vessel and within the aneurysm [5]. Nonetheless, risks of thrombosis, stent migration, and in-stent stenosis (ISS) appear or increase with SAC compared to simple coiling [6,7].
The pEGASUS stent system (Phenox) is a self-expandable, open-cell, Nitinol laser-cut stent system with hydrophilic polymer coating (HPC) surface modification for the treatment of complex intracranial aneurysms. Until now, to our knowledge there is only 1 published study concerning the use of the novel pEGASUS stent system in intracranial aneurysmal treatment [8].
We present our initial clinical experience with this novel stent system and compare available data of other intracranial stent systems.
MATERIALS AND METHODS
Patients
In a retrospective review, 12 patients with 12 complex wide-necked intracranial aneurysms were treated with a total of 12 pEGASUS stents between September 2022 and June 2023. Ten patients were treated electively while the other 2 patients harboring a subarachnoid hemorrhage (SAH) were treated in an emergency setting. Approval was obtained from the local Ethical Review Board, Aerztekammer Nordrhein, for this retrospective study (number 66/2024).
Every case was discussed in a multidisciplinary meeting with the neurosurgical team. Endovascular, neurosurgical, and conservative management were discussed with all electively treated patients.
Premedication and Anticoagulation
All 10 elective patients were provided with dual antiplatelet therapy comprising Aspirin (100 mg oral dosage [OD]) and Clopidogrel (75 mg OD) for at least 7 days prior to the procedure. Patients were only treated when adequate response to Clopidogrel (aggregation rates of <45%) and Aspirin (aggregation rates of <30%) was achieved, confirmed by aspirin reaction units and P2Y12 reaction units essays, respectively, using VerifyNow® (Accumetrics). In our cohort, there were no non-responders to Copidogrel (aggregation rates >55%) and no non- or low-responders to Aspirin (aggregation rates >50% and between 30–50%, respectively). Low-responders to Clopidogrel (aggregation rates between 45% and 55%) received 150 mg daily until adequate response was achieved.
Intraoperatively, Heparin was routinely administered in all cases with monitoring, keeping the activated clotting time between 200–300 seconds. Postoperatively, all patients were prescribed life-long Aspirin and a 12-week course of Clopidogrel.
In case of ISS on follow-up, the application of Clopidogrel was continued until the next digital subtraction angiography (DSA) control was performed.
In both cases of acute SAH, intravenous Aspirin (500 mg) and intraarterial Tirofiban (weight-adapted) were administered intraoperatively. The dose of Tirofiban included a 0.4 µg/kg/min loading dose of tirofiban for 30 minutes intraarterially followed by a 0.10-µg/kg/min maintenance infusion intravenously for 12–24 hours. After excluding SAH progression on brain computed tomography (CT) at 12–24 hours after the procedure, Clopidogrel 75 mg OD was started for at least 12 weeks combined with a life-long Aspirin 100 mg OD.
Phenox pEGASUS Stent
The pEGASUS stent system was introduced in late 2022 as the first stent system with an antithrombogenic HPC surface modification [9]. The HPC system mimics the glycocalyx, making the stent less thrombogenic in vitro. The stent is available for vessel diameters ranging from 2.5 mm to 4.5 mm and is deliverable through microcatheters with a 0.0165’’ or 0.017’’ inner diameter, with the unconstrained stent diameter available in 4 and 5 mm, while the stent length ranges from 15 to 30 mm with 5 mm increments. Each stent system features 3 radio-opaque markers at the proximal and distal ends of the stent (Fig. 1), and due to the open-cell design, the stent system is flexible and provides good anchorage, even in torturous anatomy, though resheathing and repositioning is not possible.
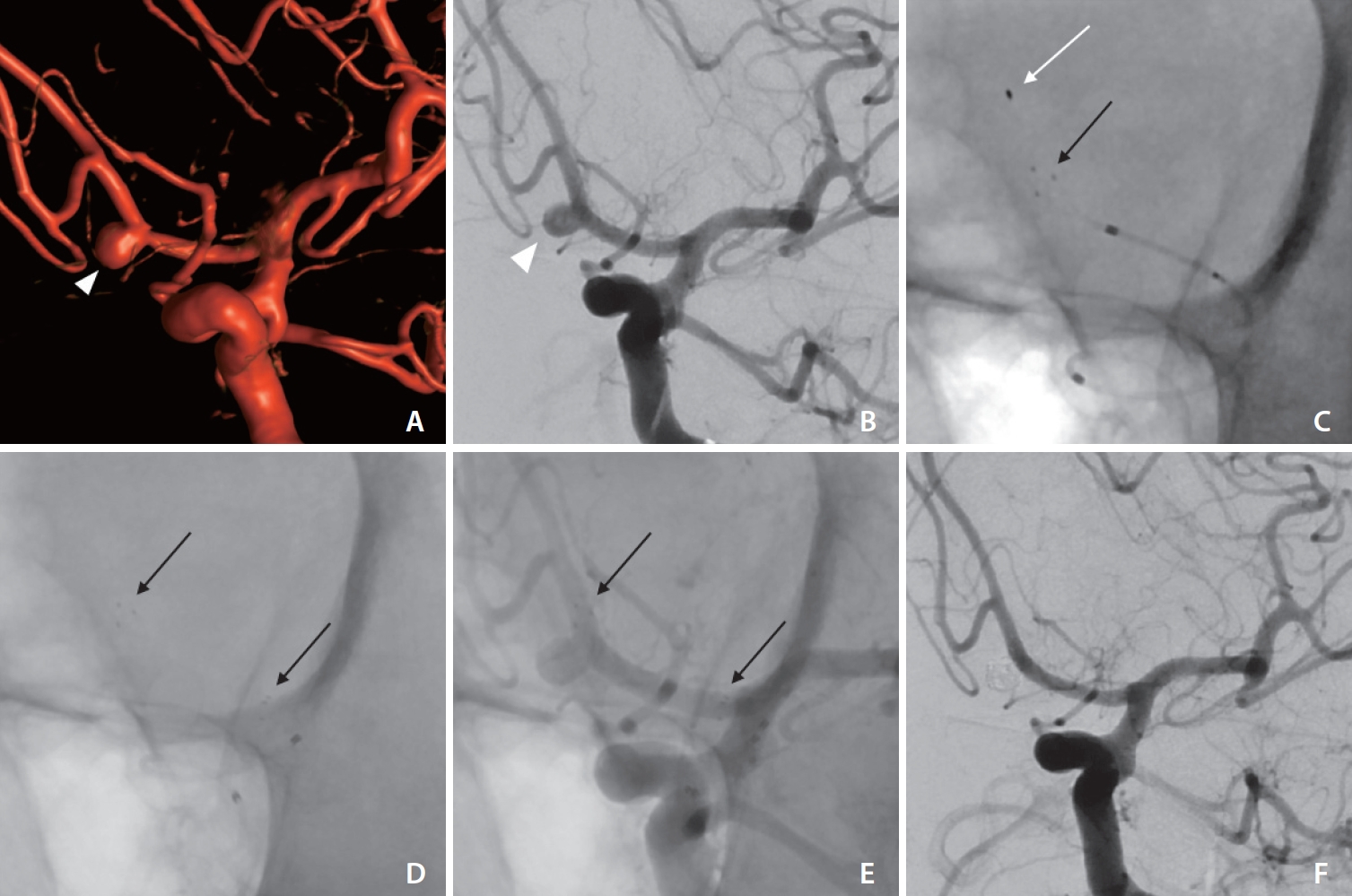
Middle aged patient with an unruptured wide-necked anterior communicating artery aneurysm (arrowheads in A, B) in 3-dimensional-angiography (A) and frontal oblique digital subtraction angiography (DSA) (B) via the left internal carotid artery. Successful placement of the pEGASUS stent (Phenox) from the left A2 into the left A1 segment (white arrow in C indicates soft distal wire tip in the left A2 during the placement of the stent; black arrows in (C–E) indicate distal and proximal stent markers). Stable complete occlusion of the aneurysm (Raymond–Roy occlusion classification I) without in-stent stenosis in DSA follow-up after 8 months (F).
Interventional Procedure
All interventional procedures were carried out on a biplane angiographic system (Artis BA Biplane; Siemens) under general anesthetic.
Our setup in all cases comprised a 6 French Envoy® guiding catheter (Codman), navigated through a 6Fr short sheath into the ipsilateral internal carotid artery (ICA) or the dominant vertebral artery depending on the location of the aneurysm. To precisely plan the intervention, 3-dimensional (3D) rotational angiography was obtained. The stent was delivered though an Excelsior SL 10 microcatheter (Stryker) in all cases. After correct placement of the stent in the parent vessel, the same microcatheter was navigated through the struts of the stent into the aneurysm and detachable coils (Axium™: 3D, Helix, and Axium™ Prime: 3D, Helix; Covidien) were used to fill the aneurysm. In 1 case, a pEGASUS stent system was used after initial placement of a Woven EndoBridge (WEB) device (MicroVention) to adjust the placement of the WEB device.
Immediately following stent deployment and aneurysm embolization, final whole-head angiograms were performed. All patients underwent a postinterventional brain CT to evaluate procedural complications.
Follow-up
Our standard follow-up protocol included a short interval follow-up at 3–6 months. Each follow-up comprises both DSA and magnetic resonance angiography (MRA) with 3D time-of-flight and contrast-enhanced MRA. The Raymond–Roy occlusion classification (RROC) was used as the standard for evaluating treated aneurysms.
A possible ISS was measured by comparing the in-stent diameter on DSA follow-up with the immediately postprocedural measured diameter. ISS was defined as ≥50% narrowing of parent artery compared to original diameter on immediate postinterventional DSA.
All examinations were evaluated by at least 2 experienced interventional neuroradiologists.
Statistical Assessment
A median test was used to compare continuous variables. Categorical variables are reported as number (%). Statistical analyses were performed using SPSS 20.0 (IBM Co.) and SAS 9.2 (SAS Institute).
RESULTS
Aneurysm Characteristics
Twelve saccular wide-necked aneurysms in 12 patients were treated using SAC or stent-assisted WEB embolization, including treatment of 2 ruptured aneurysms with SAH in the emergency setting.
There were 8 females (61.5%) patients with a mean age of 63 years (interquartile range 13). From the 10 elective patients treated with SAC or stent-assisted WEB embolization, 7 patients had no prior endovascular treatment while 3 patients presented with aneurysm recurrence after initial coil embolization.
The target aneurysms were situated at the basilar artery (BA) in 6 patients, anterior communicating artery in 3 patients, middle cerebral artery bifurcation in 2 patients, and ICA in 1 patient. The distribution of the aneurysms and aneurysmal characteristics are shown in Table 1.
Procedure
The pEGASUS stent system was deployed successfully in all patients without technical complications. There were no recorded instances of vasospasm, aneurysm perforation, or arterial dissection intra- or postoperatively. The placement of the microcatheter in the aneurysmal sac through the stent struts was feasible and performed in 11 cases (Figs. 1–3). In the other elective case (Table 2, case 11), a WEB-device was used instead of coiling of the aneurysm sac.
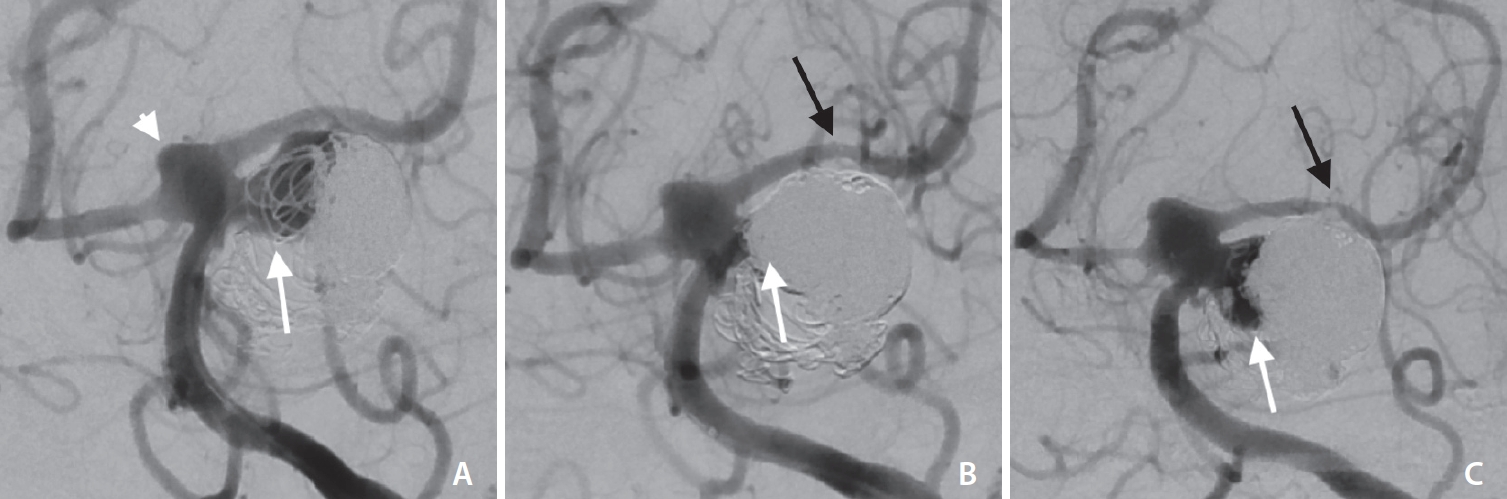
Middle aged patient with an initially ruptured and coiled basilar artery (BA) aneurysm at the left superior cerebellar artery origin. (A) Frontal oblique digital subtraction angiography via left vertebral artery shows a wide necked aneurysmal recanalization (white arrow in A). (B) After placement of a pEGASUS stent (Phenox) from the P2 segment of the left posterior cerebral artery into the BA (black arrow in B indicates distal stent markers), coiling led to an occlusion of the aneurysmal recanalization (white arrow in B). Four-month follow-up (C) shows an aneurysmal recanalization (Raymond–Roy occlusion classification IIIb, white arrow in C) and distal in-stent stenosis (black arrow in C). Note the dysplastic configuration of the basilar tip (white arrowhead in A).
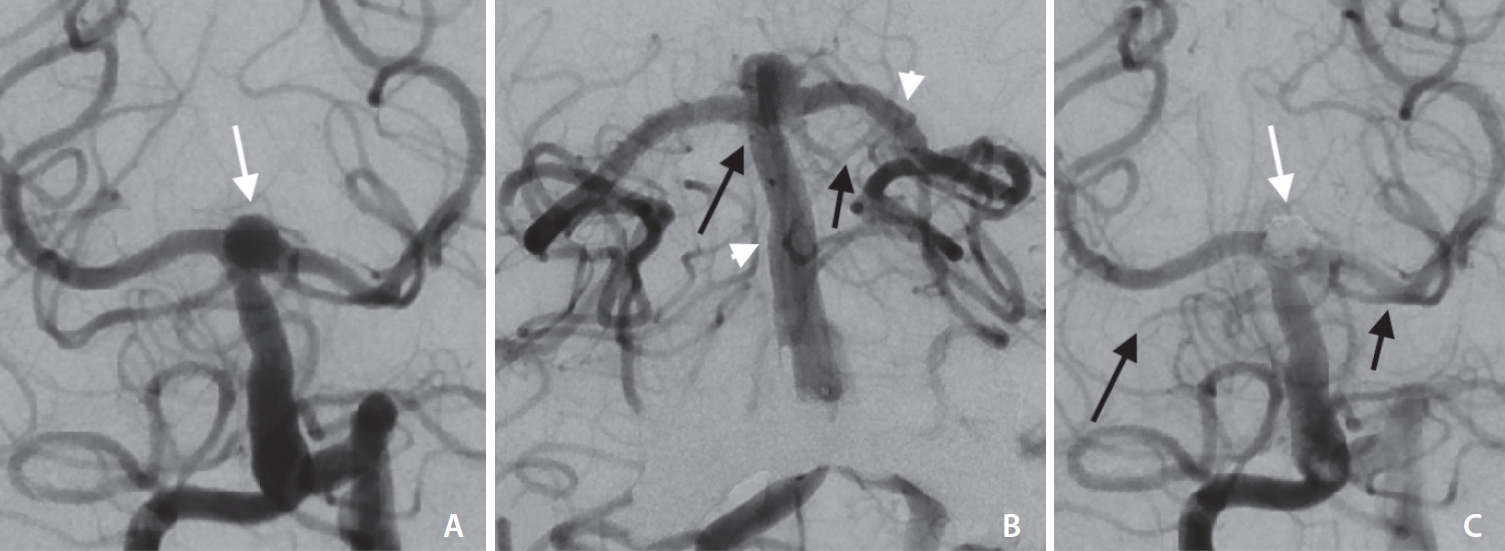
Elderly patient with an acutely ruptured wide-necked bassilar tip aneurysm (white arrow in A). Digital subtraction angiography in Towne view (A, C) and in frontal working projection for coiling after stent placement (B). After successful placement of the pEGASUS stent (Phenox) from the P2 segment of the left posterior cerebral artery into the basilar artery (white arrowheads in B), thrombi led to an occlusion of the superior cerebellar artery (SCA) on both sides (black arrows in B). With intraarterial tirofiban administration after complete aneurysmal occlusion (white arrow in C), the thrombi could be resolved in large parts with complete recanalization of the left SCA and partial recanalization of the right SCA with a remaining distal occlusion (black arrows in C).
Complications
Of all 12 cases, there was a single case (8.3%) of in-stent thrombus formation during the treatment of a ruptured basilar aneurysm (Fig. 2). The latter was resolved in with the use of intravenous Tirofiban. Follow-up brain CT showed minor bilateral superior cerebellar artery (SCA) infarctions. No other procedural complications occurred in this cohort.
Follow-up
Immediate angiographic control after SAC or stent-assisted WEB embolization (total n=12) showed complete occlusion (RROC I) in 83.3% (n=10) and near-complete occlusion (RROC II) in the remaining 2 cases (16.7%).
Of the 12 cases, 11 were available for follow-up. One patient died due to complications of the SAH not directly related to SAC. The mean follow-up time was 7.4 months (range 3–13 months).
Follow-up demonstrated complete aneurysmal occlusion (RROC) in 10 cases (90.9%; 10/11). Incomplete occlusion (RROC IIIb) on follow-up was seen in 1 case (9.1%), due to aneurysmal recanalization (initial RROC II).
One case of moderate (50%), asymptomatic ISS was seen at the distal end of the pEGASUS stent in the treatment of a BA aneurysm (Fig. 3).
DISCUSSION
Our initial experiences using the pEGASUS stent system suggests it to be a safe and feasible device combining high occlusion rates with low complication rates and low rates of ISS.
Initial postprocedural aneurysmal occlusion rates were satisfying in our cohort with complete (RROC I) or near complete (RROC II) occlusion in 83.3% (10/12) and 16.7% (2/12), respectively. This high initial occlusion rate is in line with other open-cell and closed-cell stent systems, like a study using the Accero stent with RROC I in 76.5% [10], a study using the Acandis Acclino stent with RROC I or II in 86% [11] or using the low-profile visible intraluminal support (LVIS) device jr. stent with RROC I or II in 61.7% [12].
On follow-up (mean 7.4 months), our cohort showed RROC I in 88.9% (8/9) and aneurysmal reperfusion (RROC IIIb) in only 1 case (11.1%). These rates are comparable to the rates published for other stents, e.g., the Neuroform, Enterprise, and Leo stents with a range between 73% and 87.3% for complete or near-complete occlusion [13-18]. A study by Melber et al. [19] showed slightly higher complete or near-complete occlusion using the Acandis Acclino stent system, while results of the Accero stent system showed slightly lower occlusion rates [20].
The pEGASUS-HPC stent is surface-modified with an antithrombogenic coating, which is intended to negate the need for a second antiplatelet agent. This is particularly useful when SAC is being considered in ruptured intracranial aneurysms. Despite the antithrombotic HPC surface modification, the risk of stent-related ischemic complications is not completely diminished. In the largest case series on SAC using pEGASUS-HPC stent (n=53 aneurysms), Lobsien et al. [8] reported 3 cases of stent-related ischemic changes, 1 of which was a fatal postprocedural ischemic stroke that occurred in a SAH patient who experienced stent thrombosis 13 days after treatment despite dual antiplatelet therapy. Our cohort showed only 1 procedure related complication (8.3%; 1/12). One patient harboring a ruptured BA aneurysm showed an in-stent thrombus formation in the emergency setting immediately after SAC (Fig. 3). The thrombi could be resolved with the use of Tirofiban, leading only to minor infarction in the SCA territory on both sides. The procedure related complication rate in our cohort seems comparable to the previously named studies of other stent devices in other studies. Due to the activated hemostasis of patients suffering a SAH [21], in-stent thrombus formation is a known and feared complication during the treatment of wide-necked aneurysms in the emergency setting using SAC [22]. A review by Ryu et al. [22] reported a rate of thromboembolism of 11.2% in the treatment of ruptured aneurysms using SAC. A study by Behme et al. [12] showed thrombus formation after stent placement in 6.3%.
There were no cases of vessel or aneurysmal perforation or coil migration or protrusion in our cohort. Other studies using different stent systems also demonstrate low procedural related complication rates like a study by Behme et al. [12] (LVIS) showing thrombus formation and thrombotic emboli as the only complications in 15%.
In our cohort, there was 1 case of moderate (50%), asymptomatic ISS on follow-up (11.1%, 1/9), which is in line with other open-cell and closed-cell stent systems. Here, for example, a study using the Acandis Acclino stent by Kabbasch et al. [11] showed no case of ISS, while a study by Melber et al. [19] presented ISS in 12.7% including cases of Y-stenting.
Limitations of this study include its retrospective study design. It is a single-center study with a relatively small population. The pEGASUS stent system was chosen at the experienced neurointerventionalists’ choice. The study contains elective cases of SAC as well as SAH with ruptured aneurysms treated in an emergency setting. This leads to an inhomogeneous cohort concerning procedural complication rates, clinical outcome, and anticoagulation. In total, more data in randomized controlled trials and a longer follow-up are necessary to further evaluate this novel stent system.
CONCLUSION
The pEGASUS stent system seems to be a safe and efficacious device for SAC of complex wide-necked intracranial aneurysms with high occlusion rates and low complication rates. More data is necessary to evaluate the long-term follow-up and to further compare this novel stent to other stent devices for SAC.
Acknowledgements
Frederik Boxberg has recieved travel expenses from Medtronic (Dublin, Ireland) and research grant from Acandis (Pforzheim, Germany). Katharina Schulz has recieved travel expenses from Medtronic (Dublin, Ireland) and research grant from Acandis (Pforzheim, Germany). Dominik Grieb has recieved travel expenses from Medtronic (Dublin, Ireland) and Stryker (Kalamazoo, Michigan, USA) and speaker honoraria from Medtronic (Dublin, Ireland).
Notes
Fund
None.
Ethics Statement
Approval was obtained from the local Ethical Review Board, Aerztekammer Nordrhein, for this retrospective study (number 66/2024). The study was performed in agreement with institutional guidelines and with the ethical standards according to the latest version of the Declaration of Helsinki. Due to the retrospective design of the study, a separate informed patient consent was not required.
The consent for publication is not required as the submission does not include any images or information that may identify the person.
Conflicts of Interest
The authors have no conflicts to disclose.
Author Contributions
Concept and design: FB, MSH, and DG. Analysis and interpretation: FB, MAT, KS, and DG. Data collection: FB, MSH, and DG. Writing the article: FB, MAT, and DG. Critical revision of the article: FB, MAT, KS, HL, MSH, and DG. Final approval of the article: FB, MAT, KS, HL, and DG. Statistical analysis: FB, KS, HL, and DG. Overall responsibility: FB.


