 |
 |
- Search
| Neurointervention > Volume 8(1); 2013 > Article |
|
Abstract
There are few reports describing stroke due to the acute occlusion of the vertebral artery (VA) origin successfully treated by endovascularily. The authors report a case of 78-year-old man suffering from stroke owing to acute VA origin occlusion associated with contralateral hypoplastic VA leading to basilar artery (BA) thrombosis. Cerebral angiography demonstrated that the right VA was occluded at its origin, the left VA was hypoplastic, and BA was filled with thrombus. The occlusion of VA origin was initially passed through with a microcatheter and microwire. Hereafter, angioplasty was performed followed by stenting with a coronary stent. The VA origin was successfully recanalized. Next, a microcatheter was navigated intracranially through the stent and fibrinolysis was performed for BA thrombus. The patient's symptoms gradually improved postoperatively. Stroke due to acute VA origin occlusion leading to BA thrombosis was successfully treated by angioplasty and stenting followed by intracranial fibrinolysis.
Atherosclerotic occlusive disease of the extracranial vertebral artery (VA) is a major cause of stroke and transient ischemic attack (TIA) of the posterior circulation. Atherosclerosis commonly affects the origin and proximal portion of the VA [1], and approximately one third of patients with extracranial VA stenosis are noted to have bilateral disease [2,, 3]. Occlusion or severe stenosis of the bilateral VA may cause vertebrobasilar insufficiency. Even so, there are unexpectedly only a few reports describing stroke due to an acute VA origin occlusion successfully treated endovascularily [4,, 5].
The authors report a case of stroke owing to an acute occlusion of VA origin associated with contralateral hypoplastic VA leading to basilar artery (BA) thrombosis, which was treated by angioplasty with stenting and intracranial fibrinolysis.
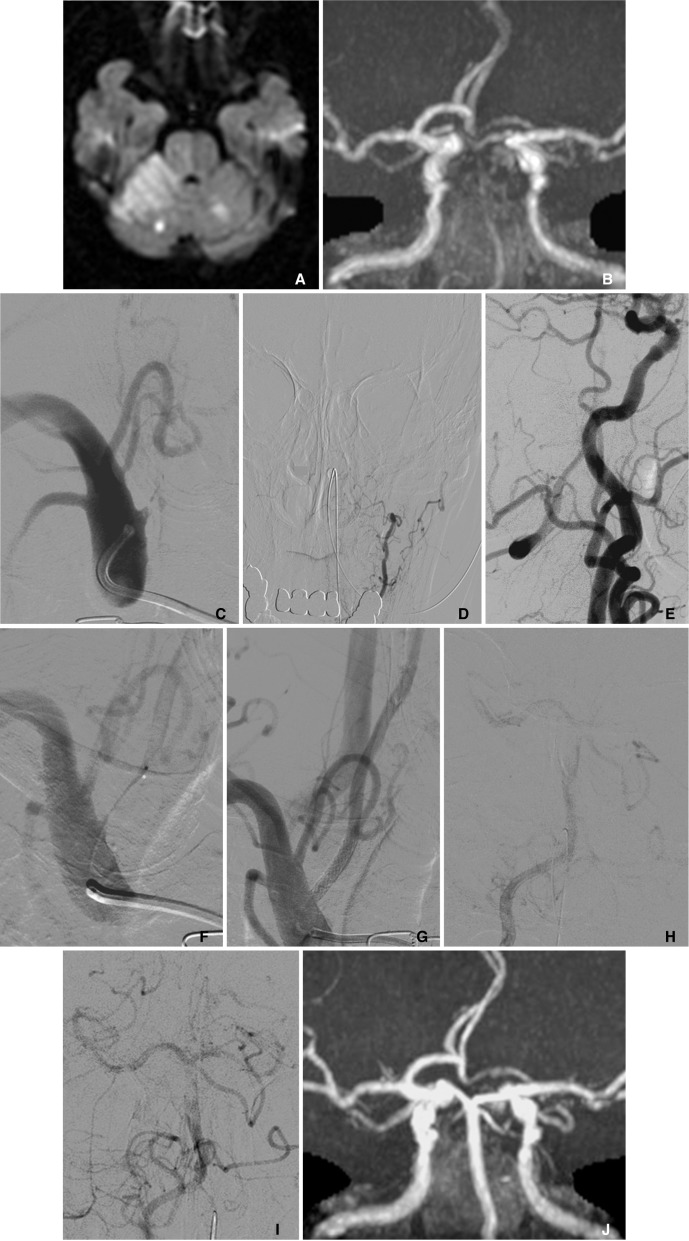
A 78-year-old man had a history of hypertension and coronary artery bypass graft for angina pectoris. However, he was not on any antiplatelet/anticoagulant medication. He was discovered after having fallen down on the street and was transferred to the hospital. He presented consciousness disturbance, left hemiparesis, dysarthria, and gaze palsy. The National Institutes of Health Stroke Scale (NIHSS) on admission was 18. MR diffusion weighted image (DWI) demonstrated a high-intensity area in the right cerebellum and brain stem (Fig. 1A). MR T2 weighted and fluid-attenuated inversion recovery (FLAIR) images showed subtle ischemic change. MR angiography (MRA) showed faint visualization in the distal part of BA and bilateral superior cerebellar arteries (SCA) (Fig. 1B).
Diagnostic cerebral angiography demonstrated that the right VA was occluded at its origin (Fig. 1C), and that the left VA was hypoplastic (Fig. 1D). The distal right VA after the midcervical portion was supplied with collateral flows via external carotid arteries and cervical arteries (Fig. 1E). The BA was filled with thrombus, although it was supplied by the antegrade flow from ipsilateral VA and the retrograde collateral flow from posterior communicating arteries. The right SCA was occluded at the origin. The left VA ended at the extracranial portion, while the left anterior inferior cerebellar artery (AICA) widely covered the left cerebellum.
The patient presented the discrepancy between the lesion on DWI and severe clinical symptom (DWI-clinical mismatch). A recombinant tissue plasminogen activator was not administered because the onset of symptoms was unknown and MR T2/FLAIR images suggested the possibility of progress of the time from onset. Therefore, endovascular intervention was immediately performed (130 minutes after the arrival at hospital).
Intervention was carried out transfemorally under systemic heparinization. A 6 Fr guiding catheter (Envoy, Cordis Neurovascular, Miami, FL, USA) was advanced to the right subclavian artery. Penetration of the occlusive site proved too difficult for the guidewire or protection devices alone Therefore, the occlusion of VA origin was initially crossed with a microcatheter (Excelsior SL-10, Stryker Neurovascular, Fremont, CA, USA) and microwire (Asahi Chikai14, Asahi Intecc, Nagoya, Aichi, Japan) (Fig. 1F). Hereafter, the microcatheter was replaced by a balloon catheter (Gateway 2.0 mm diameter and 15 mm length, Stryker Neurovascular), with catheter exchange using a long guidewire (Transcend EX floppy 300 cm length, Stryker Neurovascular). Percutaneous transluminal angioplasty (PTA) was performed followed by stenting with a coronary bare metal stent (Integrity; 3.5 mm diameter and 18 mm length, Medtronic, Minneapolis, MN, USA). Finally, VA origin was successfully recanalized (Fig. 1G). Blood was manually aspirated from the guiding catheter during and after balloon inflation to avoid distal embolism. No distal emboli were detected post-stenting. Next, a microcatheter was navigated intracranially through the stent and fibrinolysis with urokinase (total 24,000 IU) was performed for BA thrombus. Mechanical thrombectomy was not selected because of the inaccessibility. The guiding catheter was not stable in the subclavian artery and it seemed to be difficult to navigate the guiding catheter for thrombectomy through the stent placed at VA origin. Fibrinolysis was moderately effective although BA thrombus still partially remained (Fig. 1H, I). The operation was finished reluctantly because 3 hours had passed after starting intervention and approximately 5 hours after arrival at the hospital.
Dual antiplatelet therapy with 100 mg aspirin and 75 mg clopidogrel daily was administered, and anticoagulant therapy with intravenous argatroban was continued for 1 week. The patient's symptoms did not deteriorate after intervention, and all his symptoms were gradually improved postoperatively during the hospitalization. Finally, he left the hospital for rehabilitation 4 weeks later because dysarthria and left hemiparesis still remained. NIHSS at discharge was 5. And modified Rankin Scale score at 3 months was 3.
A follow-up MR image was obtained 1 week later. MR DWI disclosed a high-intensity area around the right SCA territory and the left cerebellar hemisphere mainly fed by the distal part of the left AICA. MR DWI high-intensity area around brain stem was not enlarged postoperatively in comparison with preoperative MR image. MRA demonstrated no re-occlusion of the VA origin and showed clear visualization of the right VA, BA and left SCA despite poor visualization of the distal right SCA (Fig. 1J).
Atherosclerotic occlusive disease of VA most commonly affects the origin and proximal portion of the VA [1]. Extracranial VA occlusive disease seemed to be mostly made up of benign lesions, the cause of which was attributed to: (1) the capacity to develop collateral reconstitution of the extracranial VA; (2) the usual presence of two viable arteries that join together intracranially, so that if one became compromised, the contralateral artery could compensate adequately; and (3) the slow development of luminal compromise by atherosclerotic plaques, allowing time for collateral development [6].
This article demonstrated a case of stroke due to an acute VA origin occlusion associated with hypoplastic contralateral VA, which led to BA thrombosis, successfully treated by PTA with stenting and intracranial fibrinolysis. As you may know, there were a number of reports about acute occlusion at the cervical portion of the internal carotid artery (ICA) due to atherosclerosis steno-occlusive disease. Cervical ICA occlusion often has concomitant intracranial occlusion [7], which might be brought on by distal migration of a clot from the occlusion site and/or cerebral hypoperfusion due to large vessel occlusion. Acute VA origin occlusion must be closely similar to cervical ICA occlusion. The distal migration of a clot and/or acute hemodynamic insufficiency induced thrombus at BA. In this case, contralateral VA was hypoplastic, consequently, intracranial flow of VA/BA became insufficient, and might have induced the thrombus formation of BA.
The current literature has demonstrated the safety and efficacy of PTA with stenting for VA origin stenosis. However, the cases almost all involved the non-acute period [8,, 9]. Lin DDM and colleagues reported six cases of acute-subacute posterior circulation strokes due to VA origin occlusion/near occlusion or preocclusive stenosis associated with intracranial thrombosis. In all six cases, PTA with stenting and intracranial thrombolysis was performed as in the present case. Four of six (67%) patients had excellent immediate recovery, whereas two patients expired, one of whom presented with coma at outset [5]. This result indicated that endovascular intervention might be a useful treatment option.
The concerns with the endovascular approach for this setting include two cautions. First, the difficulty in penetrating and navigating the devices across the occlusive site. It carries the risk of iatrogenic dissection or perforation of VA caused by a stray wire-tip. In the present case, a microcatheter and a microguidewire were used to raise the support and tractability for crossing the occlusion site. Second, the risk of distal migration of clots or debris into the intracranial arteries. In the present case, distal embolism did not occur, even though an embolic protection device was not used because of time limitation. However, the previous literature revealed that an embolic protection device might be useful for preventing intracranial embolisms during procedures. Iwata et al. [10] have described the use of flow reversal to prevent distal embolization during the treatment of VA origin chronic occlusion, which was similar to that reported in carotid revascularization procedures.
For the treatment of intracranial lesion, fibrinolysis is convenient regarding the distal accessibility. Mechanical thrombectomy is one of the treatment choices if the access of devices is feasible.
References
1. Hass WK, Fields WS, North RR, Kircheff II, Chase NE, Bauer RB. Joint study of extracranial arterial occlusion. II. Arteriography, techniques, sites, and complications. JAMA 1968;203:961-968 .


2. Caplan LR, Wityk RJ, Glass TA, Tapia J, Pazdera L, Chang HM, et al. New England Medical Center Posterior Circulation Registry. Ann Neurol 2004;56:389-398 .


3. Wityk RJ, Chang HM, Rosengart A, Han WC, DeWitt LD, Pessin MS, et al. Proximal extracranial vertebral artery disease in the New England Medical Center Posterior Circulation Registry. Arch Neurol 1998;55:470-478 .


4. Kikuchi O, Ushikoshi S, Kashiwazaki D, Takagawa Y, Yokoyama Y, Ajiki M, et al. Basilar artery occlusion due to embolism from the vertebral artery: three case reports. No Shinkei Geka 2010;38:669-673 .

5. Lin DDM, Gailloud P, Beauchamp NJ, Aldrich EM, Wityk RJ, Murphy KJ. Combined stent placement and thrombolysis in acute vertebrobasilar ischemic stroke. AJNR Am J Neuroradiol 2003;24:1827-1833 .


6. Fisher CM. Occlusion of the vertebral arteries causing transient basilar symptoms. Arch Neurol 1970;22:13-19 .


7. Hauck EF, Natarajan SK, Ohta H, Ogilvy CS, Hopkins LN, Siddiqui AH, et al. Emergent endovascular recanalization for cervical internal carotid artery occlusion in patients presenting with acute stroke. Neurosurgery 2011;69:899-907 .


8. Antoniou GA, Murray D, Georgiadis GS, Antoniou SA, Schiro A, Serracino-Inglott F, et al. Percutaneous transluminal angioplasty and stenting in patients with proximal vertebral artery stenosis. J Vasc Surg 2012;55:1167-1177 .


Fig.┬Ā1
MR DWI acquired on the day of symptom showed high-intensity in the right cerebellum, faint high-intensity in the right brain stem, and several small high spots (A). MRA showed faint visualization of BA and both SCAs (B). Right subclavian artery angiogram disclosed the VA origin occlusion (C). Left VA angiogram showing hypoplastic VA that ended at extracranial portion (D). Lateral view of right CCA angiogram showing that the distal right VA was supplied with collateral flow from external carotid artery system. Basilar artery was filled with thrombus although it supplied via antegrade flow from the right VA and retrograde collateral flow from posterior communicating artery (E). Right subclavian artery angiogram describing the microcatheter which crossed occlusive site (F). Right subclavian artery angiogram acquired after angioplasty and stenting showed the reconstruction of VA origin (G). Right VA angiogram, injected by lesion-crossed microcatheter, demonstrated that thrombus at BA was clearly described while right SCA was occluded (H). Right VA angiogram after fibrinolysis for BA, showing the antegrade flow of VA/BA was improved, although BA thrombus still partially remained and the right SCA was occluded (I). MRA acquired 1 week later clearly demonstrated the right VA, BA and left SCA, despite poor visualization of right SCA distal (J).
- TOOLS
-
METRICS

-
- 3 Crossref
- 3,573 View
- 56 Download





