 |
 |
- Search
| Neurointervention > Volume 15(3); 2020 > Article |
|
Abstract
Purpose
Materials and Methods
Results
Conclusion
Acknowledgments
Notes
Ethics Statement
All procedures described in this manuscript were reviewed and approved by the Institutional and Animal Care and Use Committee (IACUC) at Baylor College of Medicine prior to initiation of experiments and were completed in accordance with federal policies and guidelines.
Conflicts of Interest
Dr. Peter Kan is a consultant for Stryker Neurovascular and Medtronic. The other authors have no conflicts to disclose.
Author Contribution
Conception and design: KMC, VMS, PK, SC, and JJ. Acquisition of data: all authors. Analysis and interpretation of data: KMC, VMS, and JJ. Drafting the article: KMC, VMS, DC, and JJ. Reviewed submitted version of article: all authors. Statistical analysis: KMC. Study supervision: DC, PK, SC, and JJ.
Fig. 1.
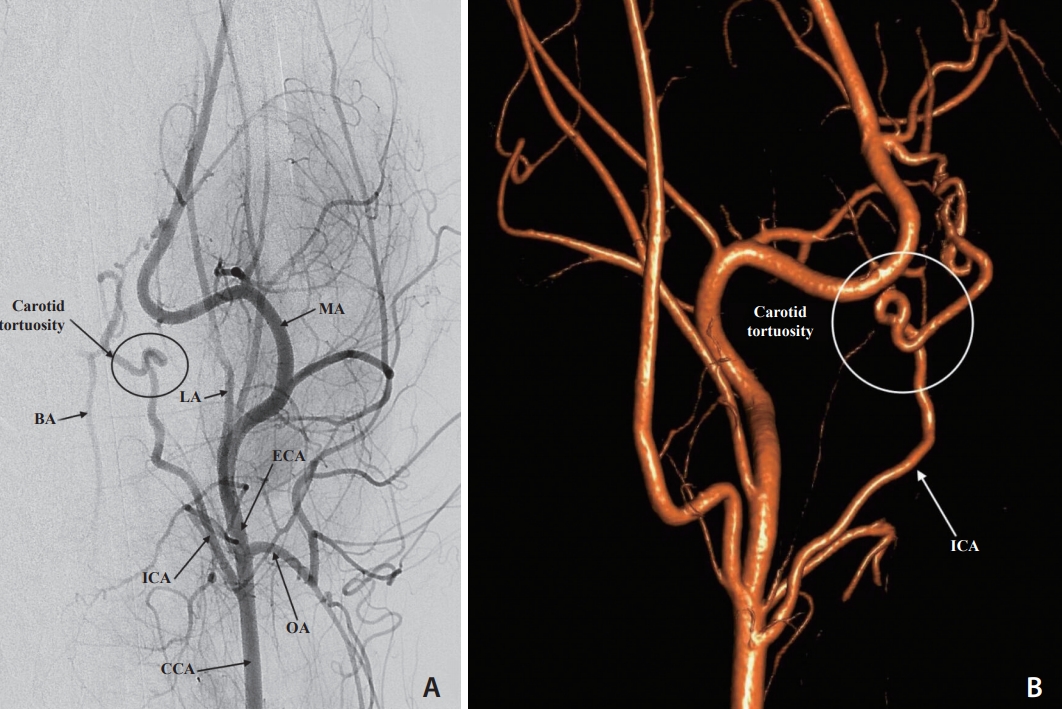
Fig. 2.
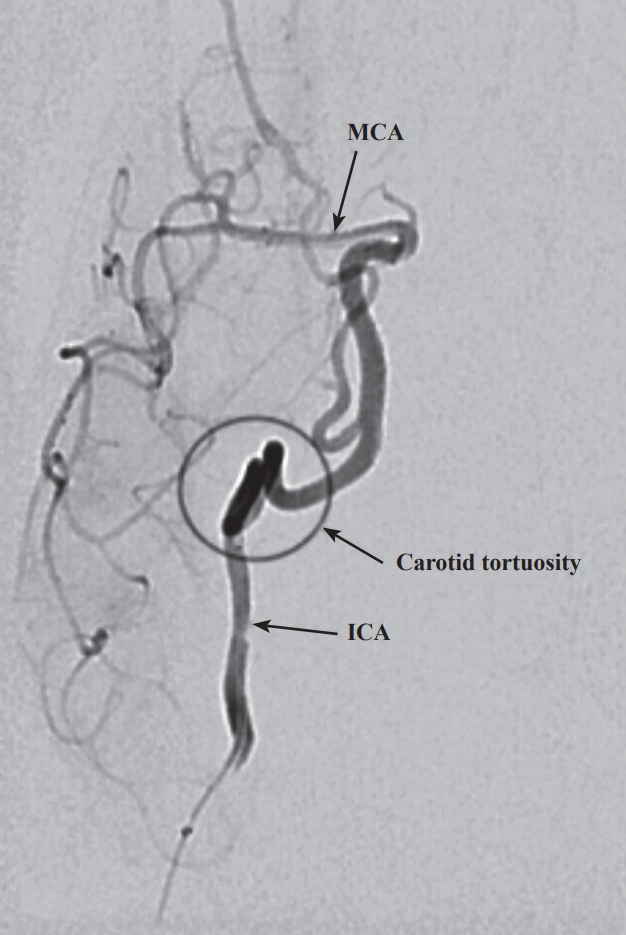
Fig. 3.

Fig. 4.

Fig. 5.
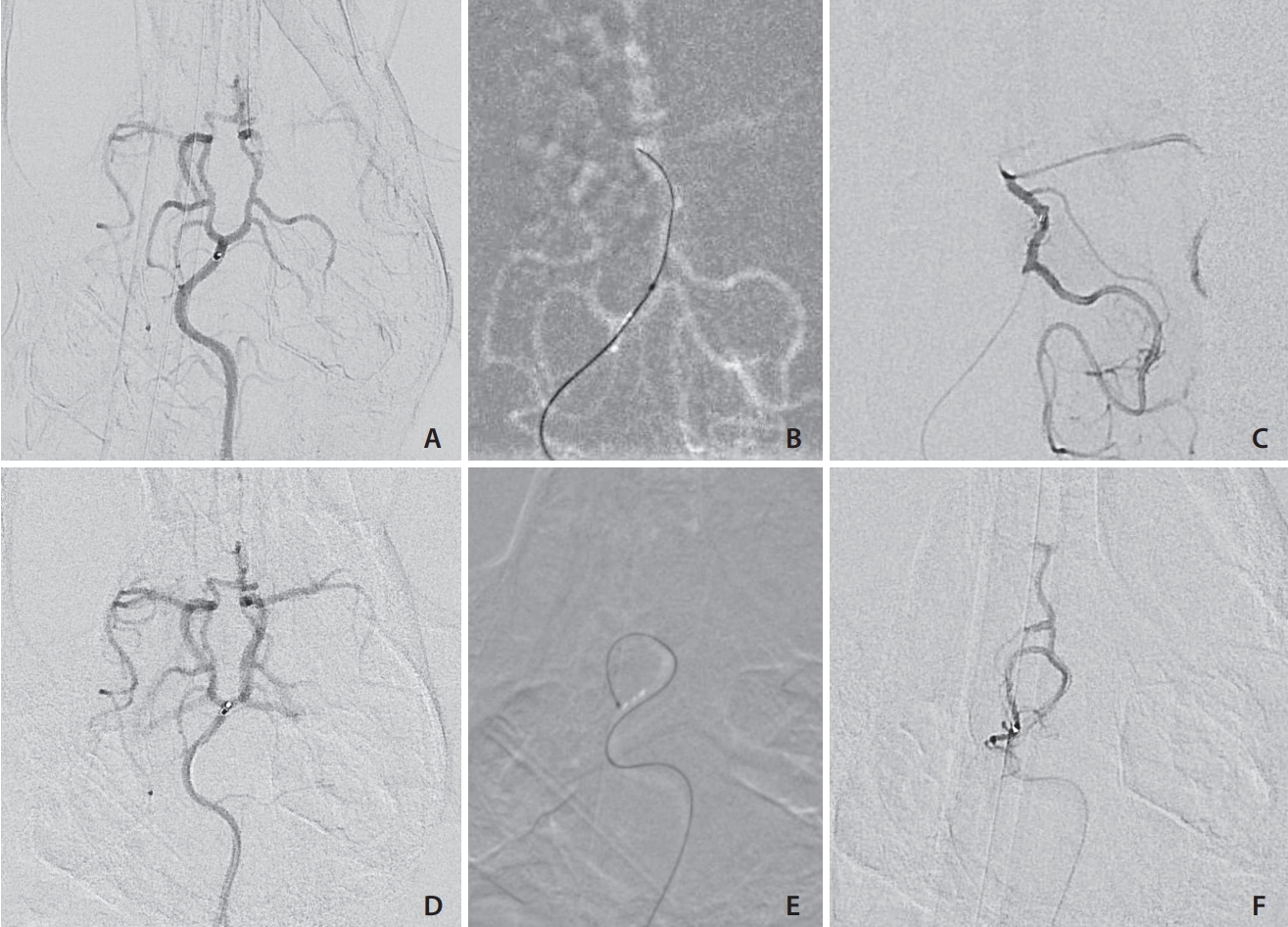
Table 1.
| Vessel | Size (mm) | Subjects measured (n) |
|---|---|---|
| FA | 4.64±0.35 | 3 |
| IA | 6.65±0.11 | 3 |
| Aorta* | 9.38±1.12 | 2 |
| CCA | 4.52±0.24 | 7 |
| ECA | 3.65±0.15 | 7 |
| ICA | 1.60±0.18 | 8 |
| BA | 1.65±0.19 | 4 |
| VA | 1.53±0.05 | 2 |
| P1 | 1.23±0.11 | 4 |
| P2 | 1.16±0.17 | 4 |
| PCOM | 1.15±0.09 | 4 |
| MCA | 1.49±0.33 | 6 |
Values are presented as mean±standard deviation or number only. Average subject weight: 30.3 kg, subject weight range: 26 to 35 kg.
FA, femoral artery; IA, iliac artery; CCA, common carotid artery; ECA, external carotid artery; ICA, internal carotid artery; BA, basilar artery; VA, vertebral artery; P1 & P2, segments of posterior cerebral artery; PCOM, posterior communicating artery; MCA, middle cerebral artery.
Table 2.
| Vessel injected | Territory visualized | Reliability |
|---|---|---|
| ICA | MCA (ipsilateral) | + + + |
| ACA (ipsilateral) | + | |
| Basilar artery | SCA (bilateral) | + + + |
| PCA (bilaterally) | + + + | |
| PCoA (bilaterally) | + + + | |
| MCA (bilaterally) | + + | |
| ACA (bilaterally) | + | |
| PCoA | PCA (ipsilateral) | + + + |
| MCA (ipsilateral) | + + + | |
| ACA (ipsilateral) | + + | |
| ACA (contralateral)† | + | |
| PCA (superselective) | PCA (ipsilateral) | + + + + |
| PCoA (ipsilateral)* | + | |
| MCA (ipsilateral)* | + |
REFERENCES
- TOOLS
-
METRICS

-
- 7 Crossref
- 5,104 View
- 197 Download






