INTRODUCTION
Long arterial sheaths are commonly used in neurovascular interventions in adults for providing increased support and the ability to deliver multiple intracranial devices [
1,
2]. Long sheaths have traditionally been avoided in pediatric neuroendovascular practice, and more so in infants and small children. In each case, the added benefits must be weighed against the larger caliber size and stiffness of these sheaths. A number of potential advantages are inherent with long sheath use which could outweigh concerns if employed safely, including the ability to co-utilize intermediate access catheters which itself affords improved technical nuances and thus safety, and potentially even shorter procedure times. In addition, dual microcatheter and microballoon techniques could be used, which would otherwise require bilateral access. Bilateral access imposes its own set of risks, not least the increased potential for loss of limb in small children. Procedural plans are devised to achieve the best possible results with available access, at times leading to a compromise in technique.
Risks entertained from using a long sheath include target vessel vasospasm or dissection from navigating a large-bore device in smaller and more reactive pediatric arteries, as well as potential occlusion and its long-term neurological sequelae. Furthermore, a long sheath employed in isolation without a shorter sheath could imply prolonged occlusion at the access site, which may be particularly undesirable during pediatric intervention. Transfemoral long sheath use was recently shown in the cardiology literature to result in a nearly 40% incidence of femoral arterial complications in infants ≤15 kg [
3]. There are no corresponding data in the neurointerventional literature.
We hypothesized that in a high volume pediatric cerebrovascular center, using dedicated protocols, the use of long sheaths for transfemoral neuroendovascular procedures can lend technical nuance without significant device-related morbidity. The goal of this study was thus to evaluate the safety and efficacy of using long vascular sheaths for neurointerventional procedures in children. A secondary objective was to look in particular at the risk profile for children ≤15 kg, who from the cardiology literature had significant complication rates with the use of long transfemoral arterial sheaths.
MATERIALS AND METHODS
An Institutional Review Board-approved retrospective evaluation was performed of transfemoral neuroendovascular procedures in children <18 years, performed using a dedicated protocol (
Table 1) in our institute over a 2-year period from 2016–2018, using long sheaths or guide catheters without an intervening short sheath. As guide catheters do not typically use a dilator, guide catheters without short sheaths were introduced over a diagnostic catheter. Long sheaths and guide catheters used were the Flexor Shuttle Sheath (5 Fr outer diameter [OD], 0.074 inch inner diameter [ID]), the Penumbra Benchmark intracranial access catheter (6 Fr OD, 0.071 inch ID), the Penumbra Neuron Max sheath (6 Fr OD, 0.088 inch ID), the Flexor Shuttle sheath (6 Fr OD, 0.087 inch ID), and the Microvention Chaperon guiding catheter (6 Fr OD, 0.071 inch ID).
A total of 27 consecutive procedures in 23 children were included, where long sheaths or guide catheters were used for intracranial access without an intervening short femoral sheath. The mean age at time of procedure was 8.4±6.3 years, range 17.0 months–16.3 years, 44% female (
Table 2). The mean weight of the children at time of procedure was 35.0±22.8 kg, range 9.8–72.2 kg. Non-neurovascular procedures were excluded. Procedural and imaging details obtained from electronic patient records and radiology picture archiving and communication system (PACS). Baseline patient characteristics were recorded, with mean age, weight, heparin dose, fluoroscopy time, and procedure time reported with standard deviations. Sex, diagnosis, procedure, reason for long sheath, procedure performed, sheath/catheter used, vessel accessed, heparin use, mean heparin dose, ultrasound guidance, complications, procedure efficacy, and post-procedural hemostasis were reported with counts and column percentages.
Values were reported as mean and standard deviation. Non-parametric chi-square test was used to compare complication rates between the two groups. P-value <0.05 was considered statistically significant. Data analysis was performed using GraphPad (GraphPad Software Inc., San Diego, CA, USA).
DISCUSSION
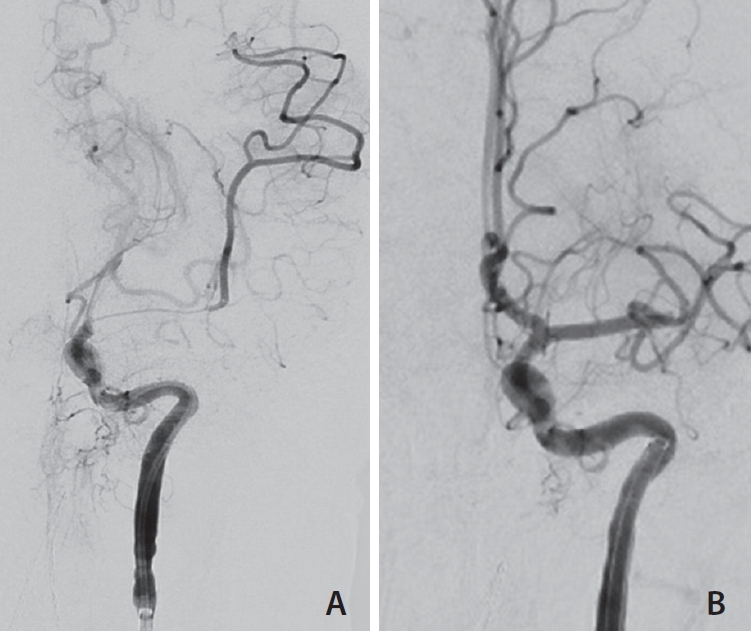
The use of long femoral sheaths in the pediatric population for neuroendovascular procedures has the potential to enable advanced techniques which would improve patient care, should the risks involved be low. At our institution, long sheath use enabled dual microcatheter techniques, microballoon assisted procedures, triaxial support (
Fig. 2A,
B), flow-reversal for mechanical thrombectomy, and intraoperative angiography with an acceptable safety and efficacy profile for these complex procedures in children. In the vast majority (59.3%) of cases, a 5 French Shuttle (Cook Medical, Bloomington, IN, USA) was used, with the various 6 Fr sheaths or guide catheters comprising the remainder. Access devices were most commonly positioned either in the cervical ICA (37.0%) or CCA (33.3%).
The proceduralist’s discretion to attempt long sheath access was typically done in the setting of angiography and embolization of arteriovenous malformations/fistulae, tumors, for intra-arterial chemotherapy or mechanical thrombectomy for stroke. Arteriovenous intracranial shunting lesions are challenging lesions, mandating careful technical nuance that is made easier by dual microcatheter technique to enable optimal nidal penetration while avoiding nontarget embolization and reflux of liquid embolic agent. Several recent technical advancements, predominantly described in the adult literature, including microballoon assistance [
4], use of newer liquid embolic agents like Onyx and precipitating hydrophobic injectable liquid (PHIL) [
5,
6], and pressure cooker technique [
7], all of which have been shown to increase the efficacy and safety of endovascular treatment, have a limited role with a single 4 Fr access that is traditionally employed in children [
8]. Similarly, treatment of pial arteriovenous fistula (AVF) and fistulous points of nidal AVMs have been thus far dependent on carefully controlled injections of n-butyl cyanoacrylate [
9-
12], which although effective, have a significant risk profile [
12]. Using long sheaths in these cases enables precise embolization
via the use of microballoons [
13,
14], or proximal coiling [
15,
16] prior to liquid embolic injection, without the need for second femoral arterial access. This not only increases the efficacy of shunt closure, but enables a degree of technical control which enhances procedural safety. In the case of intra-arterial chemotherapy, careful navigation to the ophthalmic artery is required, for targeted drug delivery. In our cohort, long sheaths were used in select cases that had dual ophthalmic arterial supply, with a prominent meningo-ophthalmic collateral resulting in competitive flow that prevented adequate antegrade perfusion in the ophthalmic artery. In these cases, long sheaths allowed the use of microballoon across the middle meningeal artery ostium to alter the local hemodynamics to maximize flow and thereby drug delivery toward the central retinal and ciliary arteries. In other cases, microballoons were used to improve microcatheter stability at the ophthalmic arterial ostium when access or distal purchase was not feasible due to angioanatomy. Improved stability of an endovascular system for the purposes of intracranial aneurysm or tumor embolization are also well-described [
17].
Mechanical thrombectomy (MT) in children poses unique technical challenges. The use of balloon guide catheters, as commonly advocated in adult MT practice, requires 8 Fr or 9 Fr femoral access, which is generally avoidable in children. Flow arrest or reversal, critical for clot aspiration and during mechanical retrieval through an intermediate catheter, can alternatively be achieved by a second operator maintaining aspiration on the guide catheter/sheath [
18,
19]. In the cases of mechanical thrombectomy in this cohort, a long sheath provided means to maintain flow arrest/reversal during clot retrieval by the ADAPT (Direct Aspiration First Pass) technique [
20]. Additionally, the use of a long sheath in these cases also facilitated direct intermediate catheter introduction into the ICA for repeat passes, which was required in 2/3 cases, and would have also permitted to easily switch to stent retriever with the Solumbra technique. An modified thrombolysis in cerebral infarction of 2b or 3 was achieved in all these cases, with one patient (162 month old, 45.5 kg) requiring intra-arterial vasodilator (verapamil) for middle cerebral artery vasospasm. It is important to note that for this particular patient, vasospasm was related to the microcatheter after aspiration, and not at the long sheath tip—thus the long sheath was unlikely to be the culprit for the complication. In the one case where a long sheath was used for intra-operative angiography in a hybrid operating room, this was inserted prior to prone positioning for a cerebellar arteriovenous malformation resection. With the sheath tip positioned in the descending thoracic aorta and the proximal portion secured to the back of the patient’s thigh, angiography could be performed in the prone position prior to surgical closure.
Unmistakably, none of the above described or proposed technical advantages would be meaningful if an unacceptable number of complications occurred or lack of success prevailed with the tools employed. Technical success was encountered in 92.6% of cases with the use of long sheaths for neuroendovascular procedures. The two “unsuccessful” cases were inability to achieve satisfactory ophthalmic arterial access for delivering chemotherapy in children with retinoblastoma. This was a result of individual anatomy, and can in fact be considered to be in spite of long sheath use than because of it. None of the patients in our cohort had vascular intimal injury directly attributable to the long sheath. One patient (2 years old) had intimal injury from the microcatheter, with focal dissection and distal thromboemboli; another (13-year old) had asymptomatic vasospasm requiring intra-arterial vasodilator therapy. Additionally, there was no increase in the rate of access-site complications. One patient (1-year old) had a groin hematoma that required a pressure dressing and prolonged admission for observation; however, no patient in our cohort had femoro-iliac occlusions, arteriovenous fistula or pseudoaneurysm. On follow-up, there were no cases of limb ischemia. This compares well with the complication rates reported from pediatric cerebral angiography performed
via small caliber short sheaths in our and other high volume centers. Hoffman et al. [
21], reporting on their experience with 309 consecutive cerebral angiograms, found a 2.9% rate of non-neurological complications (7 cases of bronchospasm, 1 transient femoral arterial occlusion and 1 groin hematoma) and no neurological complications. Similarly, Lin et al. [
22] found a 5.6% rate of non-neurological complications and 1.1% neurological complications in 697 consecutive procedures (429 diagnostic and 268 therapeutic).
We found no significant difference in complication rates between ≤15 and >15 kg groups. This is contrary to the findings reported in the literature on pediatric cardiac catheterizations. Ding et al reported arterial compromise in 11/29 (38%) infants ≤15 kg using long sheaths, as compared to 6/40 (15%) of infants with short sheaths, used for cardiac catheterization [
3]. In their cohort, time to access and sheath duration were not relevant as risk factors, but weight was, with children <5 kg being particularly susceptible to complications. Alexander et al. [
23], reporting from 486 cardiac transfemoral catheterizations in children <18 years, reported arterial compromise in 33 (6.8%) children, of whom 23 (4.7%) required treatment. Again, the smallest children, infants <6 months, were at an increased risk, and a femoral arterial diameter of <3 mm was found to be an independent predictor for loss of arterial pulse following the procedure. Similarly, arterial occlusion following pediatric cardiac catheterization was reported by Glatz et al. [
24], in 4.3% of 5,715 procedures performed in children, with smaller children and larger access catheters being implicated.
Using long sheaths without short femoral sheaths, we have shown that both neurological and access-site complications can be kept within acceptable limits while performing pediatric neuroendovascular cases including in smaller children. Although our numbers are presumably too small to draw conclusions, we provide preliminary and novel data regarding the use of long sheaths in neurointerventions in children, without an increase in complications. These results however should not be misconstrued to imply that the risks of femoral arterial occlusion and neck vessel injury are insignificant. While these comparisons are a reassuring testimonial to the use of long sheaths for pediatric neuroendovascular procedures where they add definite merit, it must be pointed out that these results originate from quaternary referral centers where patients are carefully selected, procedures performed using a stringent protocols as outlined before, and neurointerventional procedures are supported by specialized pre-, intra- and post-procedural care offered by neurosurgeons, diagnostic and interventional neuroradiologists, anesthesiologists, pediatric interventional radiologists, neurointensivists, interventional radiology technologists, and nurses. This is as much attributable to strict adherence to protocol as to the availability of multi-disciplinary expertise to preempt and manage complications. Longer term data with larger numbers is required before this can be considered as routine as in adult neurointerventional practice. As such, one should still default to using the smallest access that will permit safe and complete procedure execution.
Our study had certain limitations. Firstly, this was a retrospective sample with small numbers. However, all cases were performed adhering to a standardized protocol for pre-, intra- and post-procedural periods. Although procedures performed were heterogeneous, these are indicative of the diverse etiologies of pediatric neuroendovascular procedures and retains clinical relevance for neurosurgeons and neurointerventionists involved in the care of children with these conditions. Second, we did not include long term limb and vessel outcomes, but these are unlikely to be affected following clinical follow-up during and after discharge, and with the knowledge of pediatric angiography gained over the last many decades. Third, we do not know regarding the presence and rate of asymptomatic brain and limb emboli following an intracranial procedure, and whether these are increased with the use of long sheaths. This was not the focus of this study, and would require a well-designed forward-looking study that includes dedicated diffusion weighted magnetic resonance imaging following every procedure.