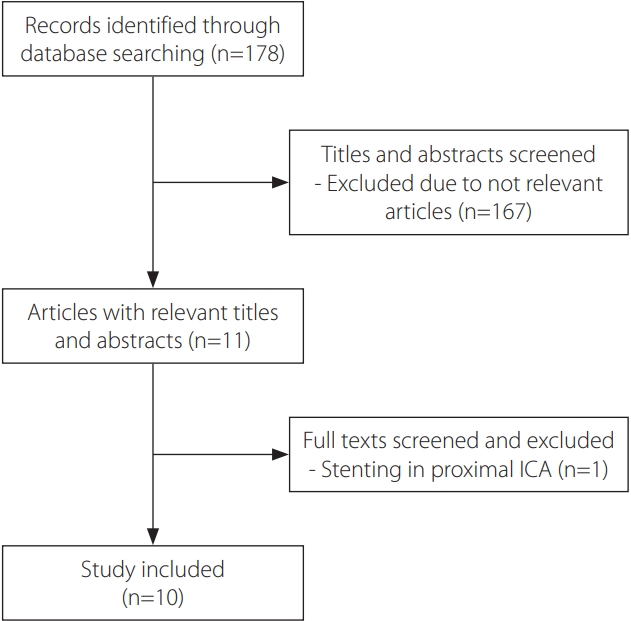
|
 |
- Search
| Neurointervention > Volume 17(3); 2022 > Article |
|
Abstract
Notes
Ethics Statement
Because the study did not involve human interaction or patient data, as institution board ethical review was deemed unnecessary. Also, consent for publication is not required as the submission does not include any images or information that may identify the person.
Table 1.
| Study | Patient # | Sex | Age (y) | Previous history | Type | Stenotic vessel |
|---|---|---|---|---|---|---|
| Kornblihtt et al. [14] (2005) | 1 | Female | 13 | Essential thrombocytosis | MMS | Left ICA |
| Rodriguez et al. [10] (2007) | 2 | Male | 37 | None | MMD | Left ICA, MCA |
| Drazin et al. [7] (2009) | 3 | Female | 40 | None | MMD | Right ICA |
| El-Hakam et al. [11] (2010) | 4 | Female | 3 | Multiple congenital disorder* | MMS | Right ICA |
| Eicker et al. [9] (2011) | 5 | Female | 18 | None | MMD | Left ICA |
| Natarajan et al. [5] (2011) | 6 | N/A | 28 | Smoking | MMS | Uni. MCA |
| 7 | N/A | Late 30s | Stroke, Smoking | MMS | Uni. MCA | |
| 8 | N/A | Late 30s | HTN, DM, HL, stroke, smoking | MMS | Uni. MCA | |
| 9 | Female | Early 30s | Oral contraceptive | MMS | Uni. MCA | |
| 10 | N/A | 51 | HTN, DM, smoking | MMS | Uni. ICA, MCA | |
| 11 | N/A | Mid 30s | HTN, DM, HL, CHF, stroke | MMS | Uni. MCA | |
| Khan et al. [6] (2011) | 12 | Female | 38 | None | MMD | Both ICA |
| 13 | Male | 39 | None | MMS | Left ICA, MCA | |
| 14 | Female | 19 | None | MMD | Left ICA | |
| 15 | Female | 32 | None | MMS | Left MCA | |
| 16 | Female | 37 | None | MMS | Left MCA | |
| Santirso et al. [13] (2012) | 17 | Female | 34 | None | MMD | Both ICA, MCA, ACA |
| Kim et al. [12] (2014) | 18 | Female | 43 | None | MMD | Left ICA |
| Argetsinger et al. [8] (2016) | 19 | Female | 55 | HTN, stroke, smoking | MMS | Left ICA, MCA |
Table 2.
| Study | Patient # | Treated vessel | Balloon angioplasty | Stent deployment | Periprocedural complication | F/U period* (mo) | Restenosis in treated vessel |
|---|---|---|---|---|---|---|---|
| Kornblihtt et al. [14] (2005) | 1 | Left ICA | No | Yes (AVE INX 3) | No | 46 | No |
| Rodriguez et al. [10] (2007) | 2 | Left ICA | Yes | No | No | 24 | No |
| Drazin et al. [7] (2009) | 3 | Right ICA | Yes | Yes (Wingspan) | No | 3 | Yes |
| El-Hakam et al. [11] (2010) | 4 | Right ICA | Yes | No | No | 0.5 | Yes |
| Eicker et al. [9] (2011) | 5 | Left ICA | No | Yes (Wingspan) | ICH, SAH | - | - |
| Natarajan et al. [5] (2011) | 6 | Uni. MCA | Yes | No | Vessel rupture | - | - |
| 7 | Uni. MCA | Yes | No | No | 12 | No | |
| 8 | Uni. MCA | No | Yes (Wingspan) | No | - | - | |
| 9 | Uni. MCA | Yes | No | No | 1 | Yes | |
| 10 | Uni. MCA | Yes | No | No | 2 | Yes | |
| 11 | Uni. MCA | Yes | No | No | 5 | Yes | |
| Khan et al. [6] (2011) | 12 | Right ICA | Yes | Yes (Wingspan) | No | 3 | Yes |
| Left ICA | Yes | Yes (Wingspan) | No | 4 | Yes | ||
| 13 | Left ICA | Yes | No | No | 2 | Yes | |
| 14 | Left ICA | Yes | Yes (Wingspan) | No | 3 | Yes | |
| 15 | Left MCA | Yes | Yes (Wingspan) | No | 2 | Yes | |
| 16 | Left MCA | Yes | Yes (Wingspan) | No | 6 | Yes | |
| Santirso et al. [13] (2012) | 17 | Left MCA | Yes | Yes (Wingspan) | No | 13 | No |
| Kim et al. [12] (2014) | 18 | Left ICA | Yes | Yes (Xience†) | No | 18 | No |
| Argetsinger et al. [8] (2016) | 19 | Left ICA | Yes | No | No | - | - |
| Left MCA | No | Yes (Wingspan) | No | - | - | ||
| Total | 11 ICA, 10 MCA | 17/21 (81.0) | 12/21 (57.1) | 2/21 (9.5) | 9.0±11.9 | 11/16 (68.8) |
Values are presented as number/total number (%) or mean±standard deviation.
F/U, follow-up; ICA, internal cerebral artery; Uni., unilateral; MCA, middle cerebral artery; ICH, intracerebral hemorrhage; SAH, subarachnoid hemorrhage.
Table 3.
| Study | Patient # | Treated vessel | Balloon angioplasty | Stent deployment | Periprocedural complication | F/U period* (mo) | Restenosis in treated vessel |
|---|---|---|---|---|---|---|---|
| Drazin et al. [7] (2009) | 3 | Right ICA | Yes | No | No | 15 | No |
| Natarajan et al. [5] (2011) | 9 | Uni. MCA | Yes | No | No | 2 | No |
| 11 | Uni. MCA | No | Yes (Wingspan) | No | 3 | Yes | |
| Uni. MCA | Yes | No | No | 4 | No | ||
| Khan et al. [6] (2011) | 12 | Right ICA | Yes | No | No | 3 | Yes |
| Left ICA | Yes | No | No | 2 | Yes | ||
| 15 | Left MCA | Yes | Yes (Wingspan) | No | 5 | Yes | |
| Left MCA | Yes | No | No | 2 | Yes | ||
| 16 | Left MCA | Yes | No | No | 2 | Yes | |
| Left MCA | Yes | No | No | 5 | Yes | ||
| Total | 3 ICA, 7 MCA | 9/10 (90.0) | 2/10 (20.0) | 0/10 (0.0) | 4.3±3.9 | 7/10 (70.0) |
Table 4.
REFERENCES
- TOOLS
-
METRICS

- Related articles in NI
-
Endovascular Treatment of Symptomatic Basilar Artery Stenosis2023 November;18(3)
Endovascular Treatment in Acute Ischemic Stroke: A Nationwide Survey in Korea2018 September;13(2)
Endovascular Treatment of Dural Sinus Malformation in Infant: A Case Report.2007 February;2(1)
Endovascular Treatment of Idiopathic Intracranial Hypertension: A Case Report.2009 February;4(1)